In ‘Comprehensive Review on Full Bone Regeneration through 3D Printing Approaches,’ the authors review new developments and solutions in tissue engineering for the formation of cells, as well as proposing an optimized temporary support geometry for treatment.
Bone regeneration continues to challenge researchers in their work as well as medical professionals attempting to improve patient treatment:
“Many research groups have been working on bone regeneration for over 10 years, but this has not led to effective therapy in a clinical setting. If it was successful, it would enhance the quality of life for millions of people and significantly reduce the absence to work due to fractures which are considered the second higher cause of working day lost,” state the authors.
“When there are fractures with a bone defect exceeding a critical size, the bone is not able to self-regenerate and, therefore, requires the use of a temporary implant (natural and/or synthetic) to serve as support and cells to help bone regeneration. In this way, tissue engineering (TE) has emerged.”
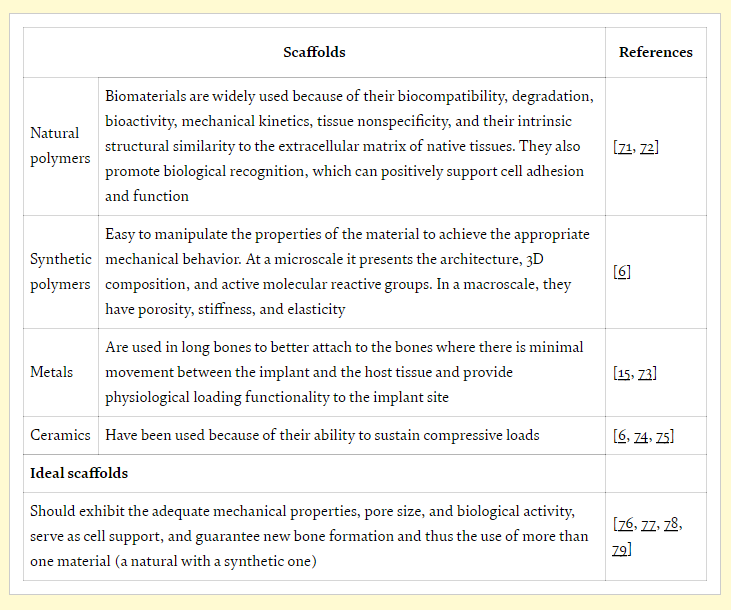
While scaffolds are used in tissue engineering for transporting nutrients and secretion of waste, the cells must be able to imitate true tissue biology, morphology, and functionality.
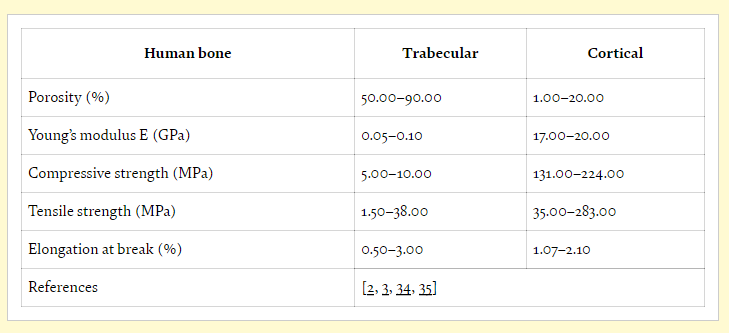
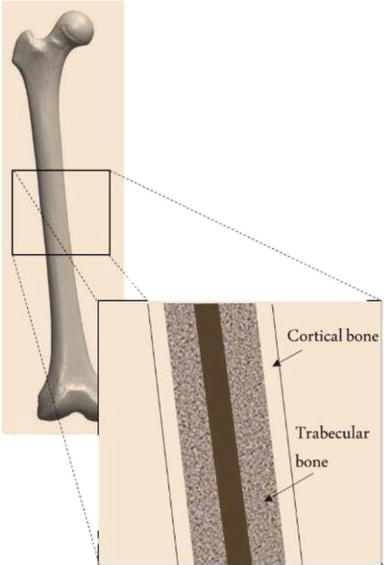
Exploring the usefulness of temporary implants, the authors state that in tissue engineering for patients, it is first critical to examine native bone tissue and mechanical properties.
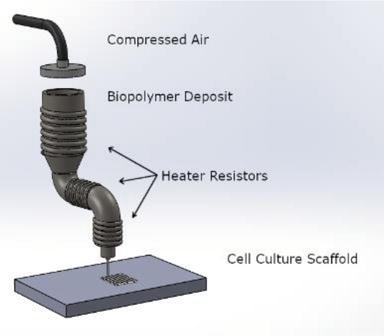
3D printed implants must be able to sustain cell viability in a secure environment, and scaffolds must possess suitable elasticity for matching regular bone. High porosity is desired in most tissue engineering, along with the use of materials that are not only biocompatible but also biologically active. During trials, animal models of fractures are often used in vivo before procedures are attempted on humans.
“Animal studies are needed to understand bone regeneration. Variables such as the amount of bone formation and its kinetics, mechanical properties and safety obtained by the scaffold, including the presence of toxic degradation in different organs and in terms of inflammatory response need to be understood in detail,” explained the researchers.
“However, bone fractures performed in animals do not represent the complexity of healing human fractures. The potential of each different type of cells both in vitro and in vivo plays here a key role.”
Even more interesting though, the authors point out that growth factors are unnecessary, with cells showing the potential to secrete optimal extracellular matrix (ECM) components.
“In vitro studies are advantageous because they offer a controlled environment to experimental test molecular and cellular hypotheses,” stated the researchers. “However, cells cultured in vitro are not replicates of their in vivo counterparts.”
While tissue engineering can be a delicate process overall in terms of working to keep cells alive, bone generation is particularly challenging—and scaffolds must be relied on to maintain the same role as tissue. Biomaterials must be able to mimic the natural environment, along with possessing identical mechanical properties of the initial bone. Appropriate levels of degradation are critical for bone regeneration, and are also dependent on corrosion resistance and materials.
Suitable materials include poly(ε-caprolactone) (PCL) or polylactic acid (PLA), both approved by the FDA and offering stability, biocompatibility, and biodegradability. Scaffolds must be osteoinductive for sustaining cells as well as being osteoconductive, providing growth. They must also serve to:
- Fill bone defect
- Ensure pore connectivity
- Encourage bone formation
- Promote bone growth
Designed in SolidWorks, the structures exhibited ‘superior advantages’ over what could be produced conventionally.
“Considering all types of materials available, associated with the desired bone regeneration and the use of synthetic polymers, as PCL or PLA, combined with collagen type I for the trabecular region and Hap for cortical region, seems to be the best strategy to follow,” concluded the researchers.
“Among the most commonly used bioreactors for bone regeneration, perfusion bioreactors appear as the most suitable, because it improves osteogenic proliferation and differentiation due to improved mass transfer and adequate shear stress. When making a design proposal for bone regeneration, it is necessary to study the mechanical effects, such as stress and tension, and link them.”
Tissue engineering continues to be an enormous area of study, from seeding human dermal fibroblasts, promoting hydrogel microenvironments, to bioprinting structures for soft tissue engineering applications.

Scaffold requirements in terms of response (left) and what should be taken into account (right) (adapted from [106]).
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Comprehensive Review on Full Bone Regeneration through 3D Printing Approaches’]
The post 3D Printing: Successful Scaffolds in Bone Regeneration appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.