3D printed bone scaffolds used for tissue engineering purposes need to have a good amount of mechanical strength, since the scaffold needs to be able to provide support for the tissue. As bone scaffolds also require the correct pore structure to help provide a good environment for the differentiation, proliferation, and repairing of damaged tissue cells, bioactive materials, such as polycaprolactone (PCL) and hydroxyapatite (HA), are needed.
 Researchers Zhiwei Jiao, Bin Luo, Shengyi Xiang, Haopeng Ma, Yuan Yu, and Weimin Yang, from the Beijing University of Chemical Technology (BUCT), published a paper, titled “3D printing of HA / PCL composite tissue engineering scaffolds,” about their work constructing nano-HA/PCL and micro-HA/PCL tissue engineering scaffolds using the melt differential FDM 3D printer they developed.
Researchers Zhiwei Jiao, Bin Luo, Shengyi Xiang, Haopeng Ma, Yuan Yu, and Weimin Yang, from the Beijing University of Chemical Technology (BUCT), published a paper, titled “3D printing of HA / PCL composite tissue engineering scaffolds,” about their work constructing nano-HA/PCL and micro-HA/PCL tissue engineering scaffolds using the melt differential FDM 3D printer they developed.
The abstract reads, “Here, the internal structure and mechanical properties of the hydroxyapatite/polycaprolactone scaffolds, prepared by fused deposition modeling (FDM) technique, were explored. Using hydroxyapatite (HA) and polycaprolactone (PCL) as raw materials, nano-HA/PCL and micro-HA/PCL that composite with 20 wt% HA were prepared by melt blending technology, and HA/PCL composite tissue engineering scaffolds were prepared by self-developed melt differential FDM 3D printer. From the observation under microscope, it was found that the prepared nano-HA/PCL and micro-HA/PCL tissue engineering scaffolds have uniformly distributed and interconnected nearly rectangular pores. By observing the cross-sectional view of the nano-HA/PCL scaffold and the micro-HA/PCL scaffold, it is known that the HA particles in the nano-HA/PCL scaffold are evenly distributed and the HA particles in the micro-HA/PCL scaffold are agglomerated, which attribute nano-HA/PCL scaffolds with higher tensile strength and flexural strength than the micro-HA/PCL scaffolds. The tensile strength and flexural strength of the nano-HA/PCL specimens were 23.29 MPa and 21.39 MPa, respectively, which were 26.0% and 33.1% higher than those of the pure PCL specimens. Therefore, the bioactive nano-HA/PCL composite scaffolds prepared by melt differential FDM 3D printers should have broader application prospects in bone tissue engineering.”
PCL is biocompatible, biodegradable, and has shape retention properties, which is why it’s often used to fabricate stents. But on the other hand, due to an insufficient amount of bioactivity, the material is not great for use in bone tissue engineering. HA, which has been used successfully as a bone substitute material, has plenty of bioactivity, which is why combining it with PCL can work for bone tissue engineering scaffolds.
“On the whole, the existing tissue engineering scaffolds preparation process have problems of low HA content, easy agglomeration, low stent strength, and single printing material,” the researchers explained.
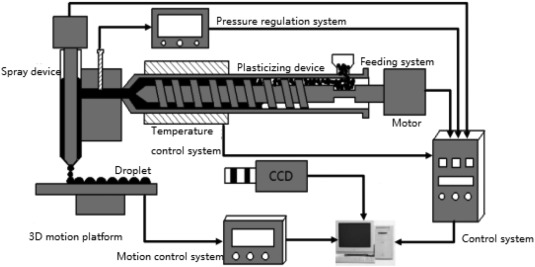
“The HA/PCL composite particles are used as printing materials, and the mechanical properties and structural characteristics of the two tissue engineering scaffolds are compared and analyzed. The raw material of the melt differential 3D printer is pellets, which eliminates the step of drawing compared to a conventional FDM type 3D printer. The 3D printer is melt-extruded with a screw, and a micro-screw is used for conveying and building pressure. At the same time, precise measurement is performed by a valve control system. This printing method shows advantages in simple preparation process of the composite material, higher degree of freedom in material selection, simple printing process, and shorter preparation cycle of tissue engineering scaffolds.”
The team mixed PCL particles and HA powder together to make the scaffolds. Their melt differential 3D printer uses pellets, and features a fixed nozzle with a platform that moves in three directions. A twin-screw extrusion granulator was used to prepare the PCL material, and the melt differential 3D printer fabricated the tissue engineering scaffolds out of the nano-HA/PCL and micro-HA/PCL composite particles.
A microcomputer-controlled electronic universal testing machine was used to test the scaffolds’ bending and tensile properties. A scanning electron microscope was used to observe the micro-HA particle size, as well as the scaffolds’ cross section, while an optical microscope was used to observe their surface structure and a transmission microscope was used to look at the nano-HA particles’ particle diameter and morphology. The scaffold material’s crystallization properties were analyzed using a differential thermal analyzer.
Testing showed that the micro-HA was spherical, with a 5–40 μm diameter, and contained some irregularly-shaped debris. The nano-HA was rod-shaped, with a 20–150 nm length.
The crystallization peak temperature of the HA/PCL composites was higher than pure PCL material, because adding HA caused its molecular chain to form a nucleate after absorbing on the HA’s surface. Additionally, adding HA to pure PCL increased the material’s melting temperature, as the latter material had crystals “of varying degrees of perfection.”
The nano-HA/PCL and micro-HA/PCL tissue engineering scaffolds “could form a pre-designed pore structure and the pores were connected to each other,” which is seen in the image below.
“…the micro-HA/PCL and the nano-HA/PCL composite tissue engineering scaffolds can form a three-dimensional pore structure with uniform distribution and approximately rectangular shape.”
These rectangular pores, with a 100-500 μm length and width, are good news for cell adhesion and proliferation, and the fact that they’re interconnected is positive for nutrient supply.
As for mechanical properties, the nano-HA/PCL specimens had the highest tensile and bending strengths – between 25 and 35% higher than the pure PCL. The micro-HA/PCL specimens had higher tensile and flexural strengths than the PCL, but the nano-HA/PCL was stronger than the micro-HA/PCL, because the HA’s modulus is higher than the PCL’s.
“In addition, nano-HA was more evenly distributed in the composite, while micro-HA had obvious agglomeration in the composite, so the tensile strength and flexural strength of nano-HA/PCL specimens were higher than that of micro-HA/PCL specimens,” the researchers wrote.
Finally, the pore structure of the nano-HA/PCL and micro-HA/PCL tissue engineering scaffolds offered a favorable environment for the discharge of cellular metabolic waste, in addition to facilitating nutrient transport and blood vessel growth. The researchers concluded that their 3D printed composite scaffolds had more potential applications in bone tissue engineering.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
The post Beijing University of Chemical Technology: 3D Printed HA/PCL Tissue Engineering Scaffolds appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.




36 Replies to “Beijing University of Chemical Technology: 3D Printed HA/PCL Tissue Engineering Scaffolds”
Comments are closed.