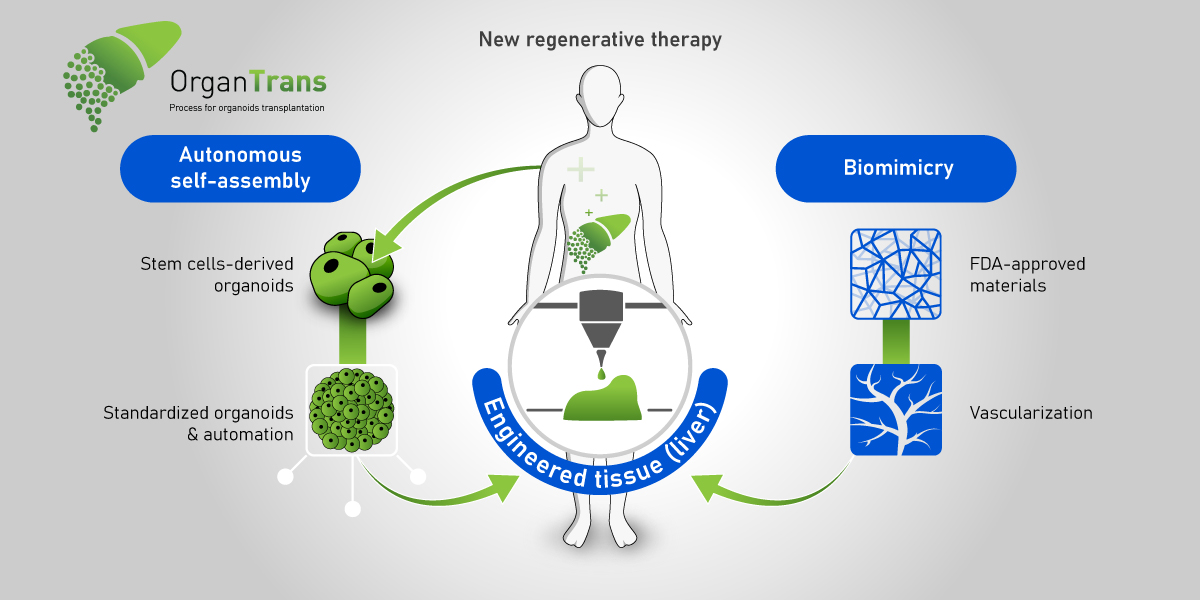
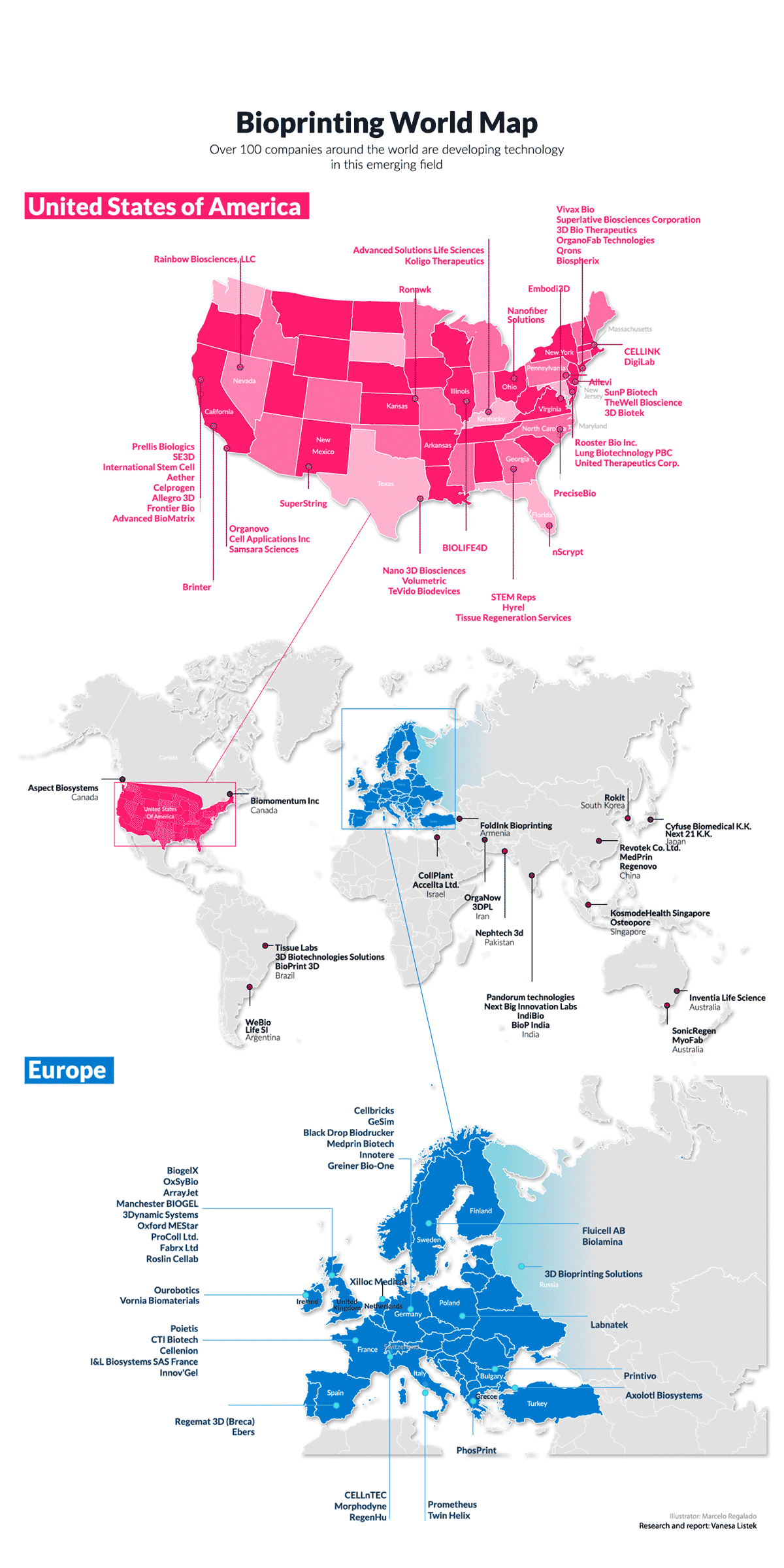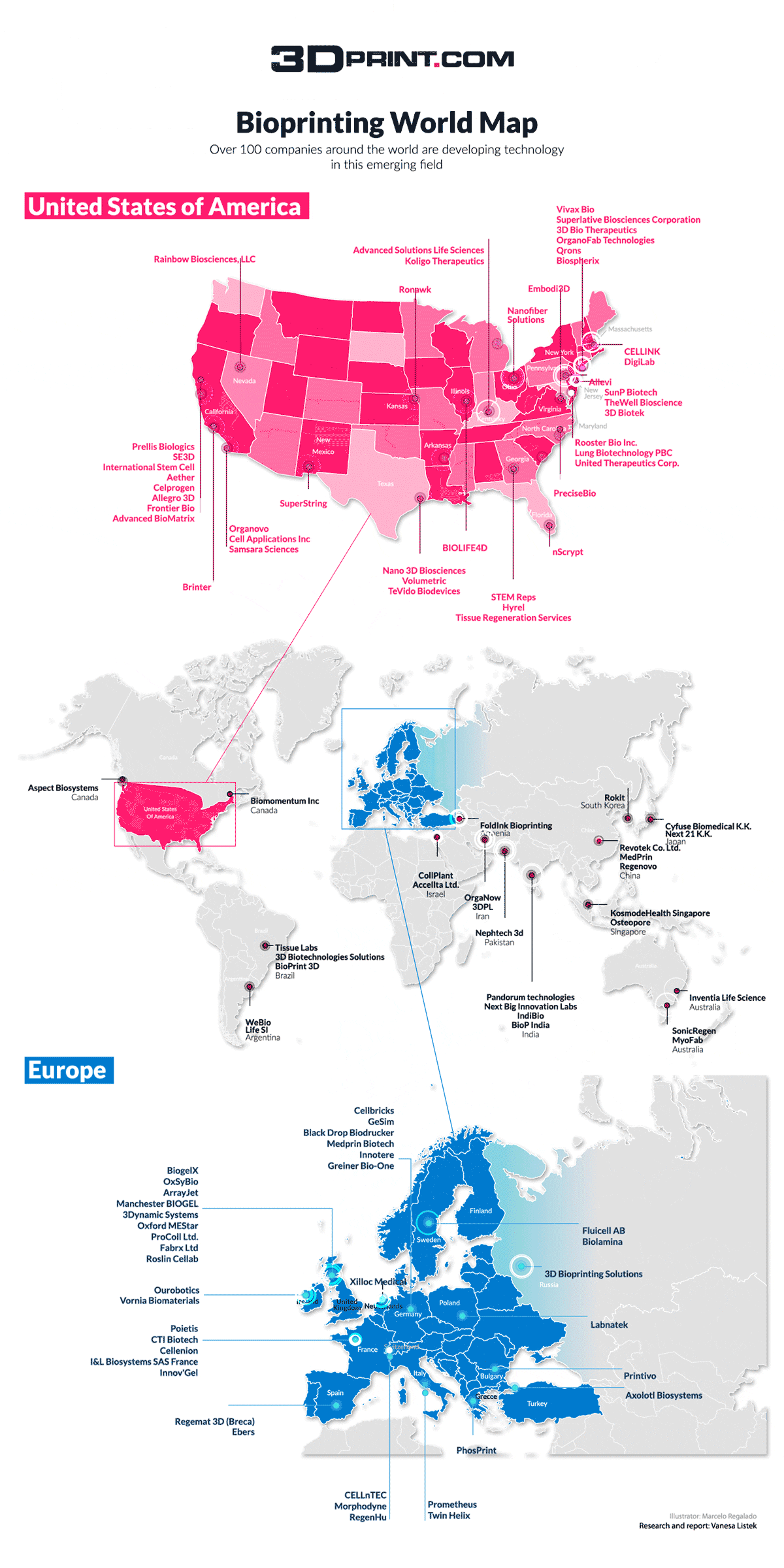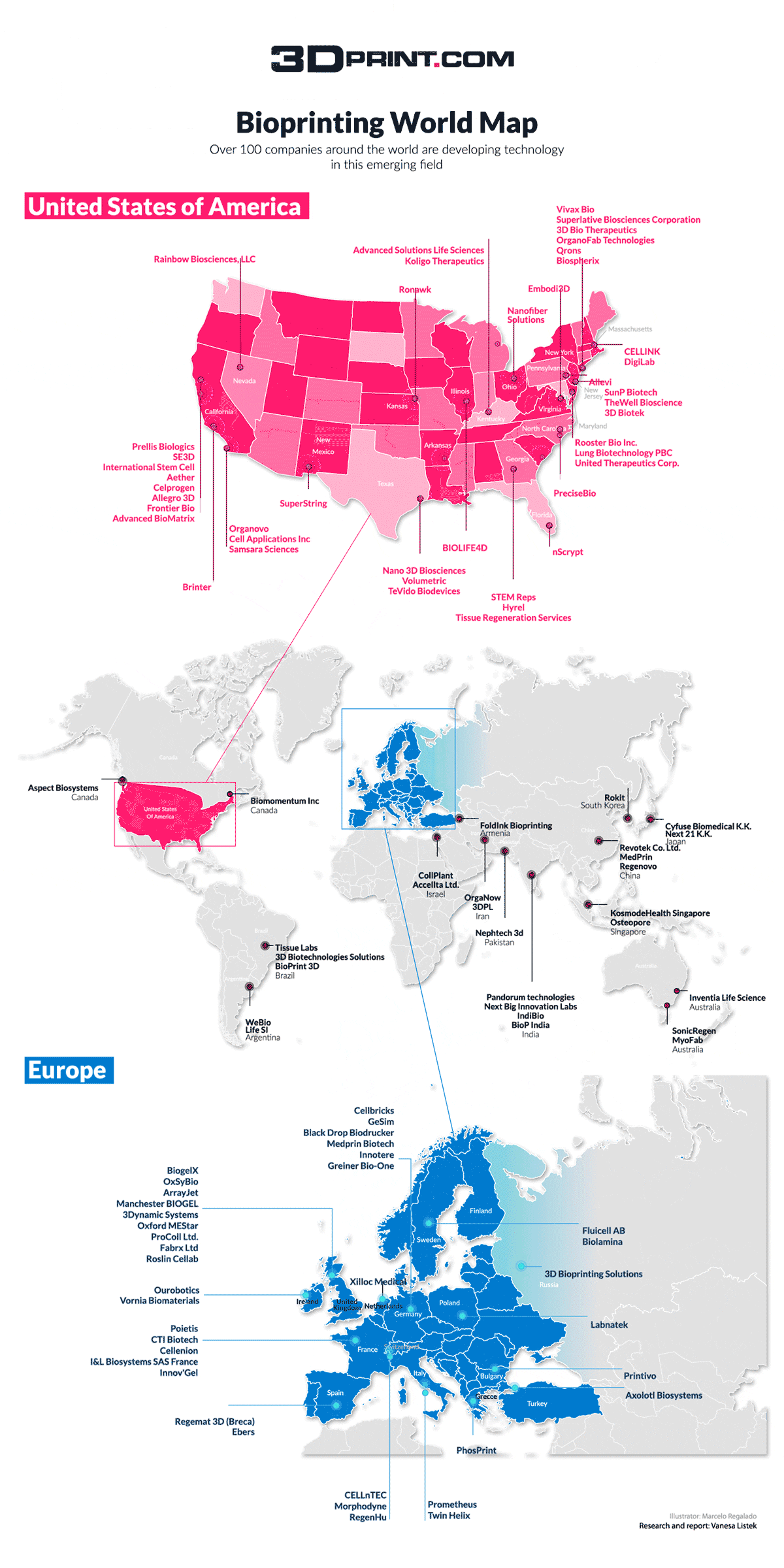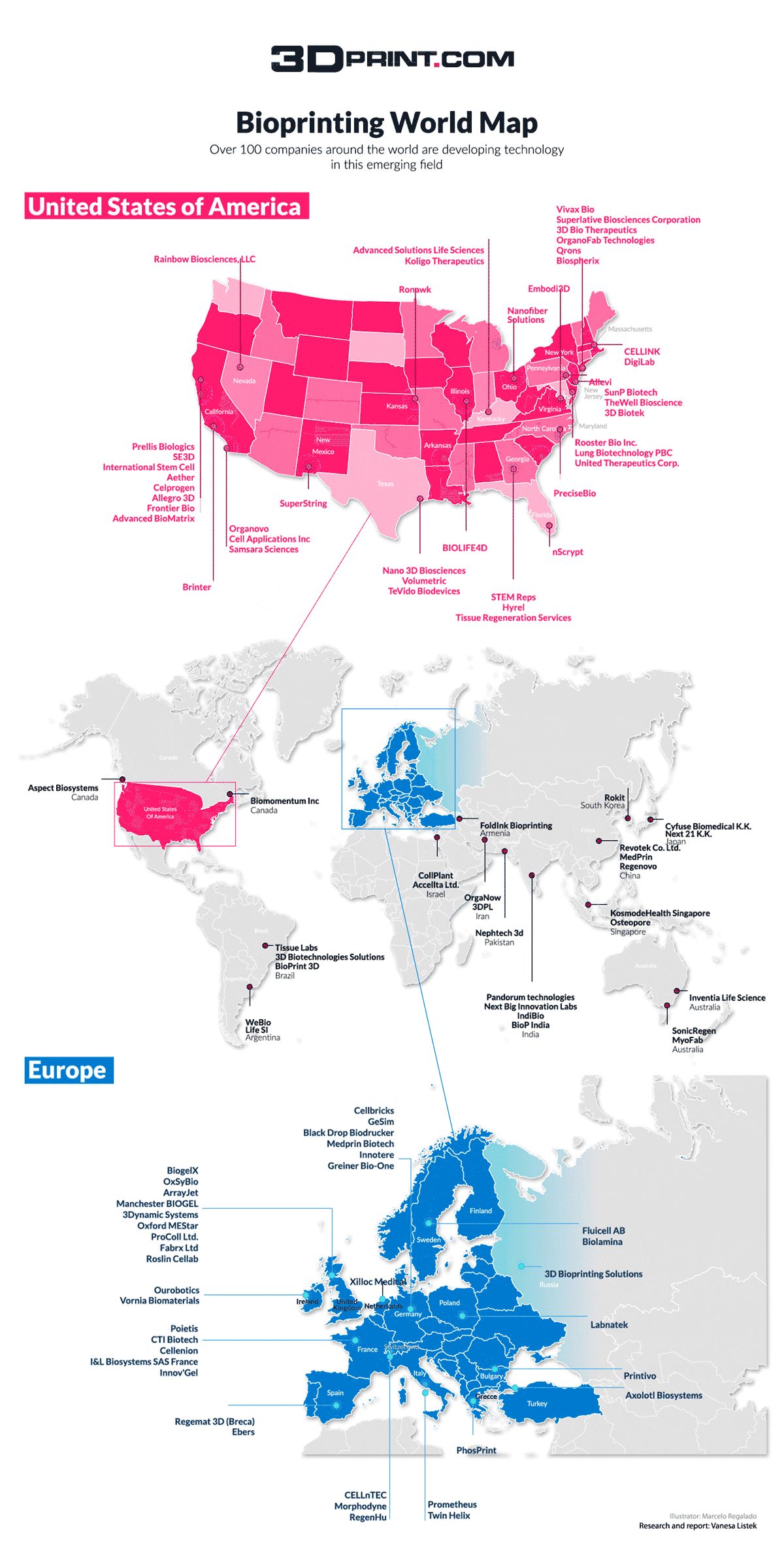
The European consortium OrganTrans is preparing to develop a tissue engineering platform capable of generating liver tissue. The proposed automated and standardized disruptive alternative solution to organ donation for patients with liver disease will stand on 3D bioprinting know-how from Swiss biomedical firm regenHU. Coordinated by Swiss research and development center CSEM, the eight partners and two transplantation centers engaged in the consortium will be using regenHU’s 3D bioprinters to produce organoid-based liver constructs with organoid laden bioinks.
In April 2020, we reported that OrganTrans would tackle the important healthcare challenge of end-stage liver disease (ESLD) by capitalizing on advancements in the regenerative medicine field, like using biofabricated liver tissue, to develop an entire value chain from the cell source to tissue engineering, biofabrication, post-processing and testing, and liver transplantation under the “compassionate use exemption” regulation (which provides an important pathway for patients with life-threatening conditions to gain access to unproven human cells and tissue products). To understand the key role of biofabrication in this innovative project, 3DPrint.com asked regenHU’s new CEO, Simon MacKenzie, to tell us more about the challenges that lie ahead for the European consortium and his company.
The project officially began in January 2020, what can we expect when it ends in December 2022?
The current goal of this project is to create a functional biofabricated liver construct that can be implanted into a mouse model. I consider that the OrganTrans team will accelerate new solutions for patients with liver failure. It is challenging, but we do envision successful in vivo trials. Of course, this major achievement will not be the end of the story; significant work and research will still be required to transfer these results to human clinical trials. The major remaining challenges will probably be the process scale-up to produce larger tissue and regulatory aspects.
Will this research be groundbreaking to treat liver disease in the future?
Demonstrating the feasibility of the approach in a mouse model will be groundbreaking for the disease because it will demonstrate its potential as an alternative to transplantation. Diseases like NASH [nonalcoholic steatohepatitis, an aggressive form of fatty liver disease] are increasing dramatically, and likely to be a leading cause of death within the next few years. Moreover, the difficulty of detecting the disease until it is potentially too late leads to significant challenges for therapeutic intervention, meaning transplantation will remain the main option for severely affected patients. This well-recognized need, along with the lack of donor organs will ensure bioprinted livers will continue to be well funded. But the value of the project goes beyond liver disease, as the new technologies developed in the frame of OrganTrans will not be limited to liver applications. They relate to the challenges of biofabrication of any organoid-based tissue, which can potentially be beneficial for a large variety of indications.
Can you tell me more about the role of regenHU within the OrganTrans consortium?
Such a complex and ambitious endeavor needs very different and complementary knowledge and competences. Teamwork will be a central element, first to enable, then to accelerate, these new solutions. With this in mind, we have been reorganizing regenHU to bring better project collaborative capabilities to this project, and others like it that we are engaged in. regenHU is a pioneer and global leader in tissue and organ printing technologies converging digital manufacturing, biomaterials, and biotechnology to lead transformational innovations in healthcare. We focus on delivering advancements in the instruments and software required for tissue engineering, and our technology evolving along with the biological research of our partners. We, therefore, consider these partnerships with the scientific community critical for our development.
regenHU is one of the largest contributors to this project, is this part of the company’s commitment to regenerative medicine?
We can see the need for biotechnology solutions for a wide range of disease states. Our strengths are in engineering the instruments and software necessary to allow the producers of biomaterials and the suppliers of cells to combine their products to achieve functional tissues and organs. Our commitment is to provide disruptive technologies that will enable the community to make regenerative medicine a reality, with precision and reproducibility in mind, for today’s researchers and tomorrow’s industrial biofabrication needs. One of the key challenges is the current limitation in the scale and volume of bioprinting which is linked to the reproducibility of the print. To progress into the manufacture of medical products, bioprinters will need to operate at a scale beyond current capabilities. We design our instruments with these goals in mind and have assembled a team to solve the many challenges to achieve this.
How advanced is the bioprinting community in Europe?
The 3D bioprinting field is several years behind mainstream 3D printing, with the industrialization of the instruments, biomaterials, and cells required before bioprinting can progress to commercial-scale biofabrication. However, as with continued development seen in 3D printing, the technology convergence required for tissue and organ printing that changes medical treatments will become a reality through the efforts of engineering companies like regenHU, biomaterial developers, and human cell expansion technologies, being combined in projects such as OrganTrans.
As the newly appointed CEO of the company, how do you feel taking on this project?
Successfully entering the OrganTrans consortium is just one part of the company. regenHU investors see my arrival as the catalyst to bring regenHU to the next stage in its evolution. Our goal remains the production of industrial biofabrication instruments capable of delivering the medical potential of bioprinting, novel bioinks, and stem cells. To achieve this, we are enhancing the team and structure of the company, bringing forward the development of new technologies and increasing our global footprint to better support our collaborative partners. I have spent many years in regenerative medicine and pharma and can see the potential of bioprinting to revolutionize many areas of medical science, so joining regenHU was an easy choice. As CEO, my main role is to provide the right support structure to enable our entrepreneurial engineering teams to thrive and be brave enough to push boundaries. Additionally, as we cannot achieve our end goal on our own, I am here to nurture the important connections with our user community. Only by listening to their valuable insights and solving problems with them, we will push the technology onward.
The post regenHU CEO: Bioprinting Will Strengthen OrganTrans Project to 3D Print Liver Organoid appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.