TaylorMade Uses Formlabs to Prototype Better Golf Clubs with 3D Printing
Golf company TaylorMade extensively used Formlabs machines to prototype a better golf club. The company’s work illustrates a few key emerging trends: that desktop machines could partially displace services, that desktop machines could be used instead of more expensive in-house systems, and that vat polymerization can be used for more functional prototypes than in the past.

The TaylorMade team wanted to look at the weight distribution in the club head of its new Sim Fairway club. They wanted to “lower the center of gravity, improve turf interaction, and create a more forgiving face.” I’m entirely unsure what the last two terms mean but I love them already. They do just go to show you that, for different markets and products, wholly specific engineering terms and goals can come to center stage. For the team prototyping, 3D printing allows them to see and feel the weight distribution. They used Formlabs Draft Resin for initial parts and then turned to Grey Resin for later prototypes. One benefit that TaylorMade enjoyed was the fast turn around times, the other comparatively low cost, and the ability to combine separate components into assemblies.
By printing parts separately and mating them, the team was able to make full assemblies of all of the various shapes of weighted sole plates. TaylorMade’s Chris Rollins said that “the way the Grey Resin parts would mate together, the resolution of the parts, was something we could not find in many other printers, We had better results by printing parts separately and then combining them together.”

Whereas most service bureaus still consider desktop 3D printers mere toys, we cannot ignore that they are improving. Desktop machines are usually significantly lower cost than service bureau parts. Service bureaus, of course, give you a much broader selection of materials and technologies to work with. A desktop 3D printer will never satisfy all of a large company’s prototyping needs. You’d need several machines with a wide variety of materials and people with expertise to make many different types of parts. Even then, the sheer amount of labor involved in 3D printing and finishing parts means that there will have to be in-house resources available to sand and remove supports from parts. Post-processing costs of 3D printed parts are significant and often not fully taken into account.
Most companies will, therefore, use in house desktop systems for initial prints at the engineer’s desk and turn to services for later functional testing or visual prototypes for photography. Desk-side prototypes are always handy and useful to get the discussion going, but, if you require 100 prototypes that need to fit and be available to test a door handle, it usually won’t work with desktop machines or a quick cost calculation may tell you that the in-house parts could indeed be very expensive for you, with all of the prep and post-processing time.
If it would take you ten minutes per part to do file prep, wash, flash, remove supports, and sand, then that would be 16 hours for 100 parts that would set you back 560 Euro in labor (if you assume its a skilled office worker whose total cost would be 35 per hour). Formlabs actually has a super nifty ROI calculator that lets you play with these numbers. On the whole, desktop machines don’t always make sense, but when they do, they convey significant cost advantages and speed up engineering teams. Many machines worldwide sit idle for want of a CAD designer or new nozzle. To me, the only logical response that services could have is to offer to maintain, service, and support fleets of desktop 3D printers at company locations.

At the same time, talk of mating parts may have some other players in the market worried about their prospects. Higher-end vat polymerization machines and those based on other 3D printing processes could also be displaced by these kinds of systems. In-house systems like PolyJet ruled the high-definition, in-house prototyping world. Now capable of color, these systems are still very formidable, especially for visual prototypes. They’re also more reliable and generally a bit easier to use.
I don’t think that someone is really going to make a decision between a J55 or a J750 and a Formlabs machine just yet. But for many edge cases, it could be an interesting choice. In many instances, you can get five or 50 Form 3’s for the cost of your larger industrial system. If you need large parts, then you must pick the larger machine, but, if you don’t, then a cheaper up-front cost is tempting.
The resin costs of Formlabs systems are significantly less than those of larger industrial systems. And here is where the choice goes from tempting to logical, if you need a lot of small parts, the savings will be significant in the long run. If consumables costs on industrial and prototyping systems remain too high, then lower-cost systems will keep making inroads into the office. In particular, companies new to 3D printing may want to start off with a much cheaper system to get their feet wet. If the Formlabs machines suffice, these firms may never again consider a higher-end machine.
Resins and photopolymers are problematic, from a safety, environment, and cost perspective when compared to many thermoplastics. They also lacked strength, UV resistance, and HDT. A few years ago, only very few firms considered vat polymerization for the prototyping of functional products or form and fit. Improvements in photopolymer chemistry have, however, meant that heat deflection and strength are improving. With high resolution, this may mean that vat polymerization machines could occupy a larger share of the prototyping market.
Still less durable and tough and with higher part costs, due to material cost and support removal, FDM still will be a better choice for most (even with the uglier parts). On the whole, we can see competition between technologies and between machines at very different price points. Services will feel desktop machine growth. Overall, we’re seeing blended usage patterns emerge where companies use many technologies from different vendors to get their 3D printing done.
The post TaylorMade Uses Formlabs to Prototype Better Golf Clubs with 3D Printing appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
3D Printing Webinar and Virtual Event Roundup, July 7, 2020
We’ve got plenty of 3D printing webinars and virtual events to tell you about for this coming week, starting with nScrypt’s webinar today. 3Ding and Formlabs will each hold a webinar tomorrow, July 8th, and 3D Systems is hosting a virtual event on the 8th. There are two more webinars on July 9th, by KEX Knowledge Exchange and ASME, and Additive Industries is holding a virtual event that day. Finally, a 3D Health Hackathon will take place starting July 10th.
nScrypt’s Cutting Edge of Digital Manufacturing Webinar
On June 30th, nScrypt held the first of a two-part Cutting Edge Digital Manufacturing webinar series, and is holding the second part today, July 7th, at 1 pm ET. In part two of “Pushing the Envelope of Digital Manufacturing,” the speakers will be Eric D. Wachsman, PhD, from the University of Maryland; Eduardo Rojas, PhD, with Embry-Riddle Aeronautical University; Hjalti Sigmarsson, PhD, from Oklahoma University; and Craig Armiento, PhD, with the University of Massachusetts Lowell.
Topics of discussion in this webinar include the use of metamaterials, building radio frequency devices, systems, and the first 3D/volumetric electrical circuits and antennas, and the state of the art of 3D manufacturing. Register here.
3DIng “Let’s Talk 3D Printing” Webinar
Indian 3D printer manufacturer 3Ding recently began holding a weekly webinar about 3D printing-related topics, such as SketchUp training, different types of 3D printing, OpenSCAD, slicing, applications in rapid prototyping, and how to choose a 3D printer. Tomorrow, July 8th, the topic of the weekly webinar will be “Live Demo of FabX, Hydra Series 3D Printers & AMA.”
Surendranath Reddy, the founder, CEO, and CTO of 3Ding, is leading the remote webinar session, which will take place at 6:30 am ET and last about 45 minutes. You can join the session here.
Webinar on Formlabs’ New Materials
Formlabs recently launched two new materials, Flexible 80A and Elastic 50A resins, which allows customers to make soft, flexible parts with ease. In a webinar on July 8th at 2:00 pm ET, attendees will get to learn all about these resins with the company’s Materials Product Manager Kathy But and webinar specialist Faris Sheikh. Topics will include when to use these materials, optimal applications, 3D printing material properties like spring back, tensile strength, and shore durometer, and the Ross Flex Test.
“To make soft and flexible parts with traditional methods, such as RTV moldmaking, can be a lengthy process. If you’ve also tried directly 3D printing flexible parts, you probably know there’s not many high performing materials available. That is now changing.
“With the launch of our Flexible 80A and Elastic 50A Resins, you’ll be able to easily fabricate flexible parts that are both soft and hard.”
Register here.
3D Systems’s Virtual Tradeshow
3D Systems is holding a virtual event on July 8th in order to teach attendees how to transform their manufacturing workflows. There will be a keynote address, networking opportunities, multiple live webinars, and even a virtual exhibition hall. The company will provide examples of digital manufacturing solution workflows with plastic and metal additive manufacturing, subtractive manufacturing, and on-demand services.
“Businesses are focused on lowering risk, resolving supply chain dependencies, streamlining supplier distribution and avoiding interruptions to supply access.
“Join 3D Systems at this exclusive virtual event to find out how Digital Manufacturing Solutions designed for today’s production needs, enable you to integrate additive and subtractive technologies into your manufacturing environment and workflow — providing increased agility, quicker lead times, improved productivity, and allowing you to offer new innovations to your customers.”
All presentations will be in English, and available on-demand for 30 days. Register here.
KEX Knowledge Exchange on Powder Bed Fusion
KEX Knowledge Exchange AG, a former spinoff of Fraunhofer IPT, offers technology consulting. As a service to its industrial and research partners, the company also has a web platform that offers over 7,000 profiles of AM technologies and materials, in addition to industry news, and has now launched a section devoted to webinars, with topics including post-processing and powder bed fusion (PBF) 3D printing.
“Together with one of our appreciated network partners, the ACAM Aachen Center for Additive Manufacturing, we now launched a webinar section,” Jun Kim Doering, a technical writer with KEX, told 3DPrint.com. “Due to the COVID19 situation, ACAM has shifted their focus to an online offering, including webinars on different aspects of the AM technologies and applications.”
The first, “Webinar Powder Bed Fusion (PBF) – Advanced insights into Process, Parameters & Hardware,” will take place this Thursday, July 9th, and Erik Feldbaum, ACAM Aachen Center for Additive Manufacturing, will speak. It’s free for ACAM members, and will cost €175 for non-members.
ASME on 3D Printing in Hospitals
AM Medical, powered by ASME International, will be holding a free, live webinar this Thursday, July 9th, on “Building the Business Case for 3D Printing in Hospitals.” Point-of-care manufacturing leaders will discuss necessary skills, where to find the proper resources, how to address reimbursement, and other important questions during the hourlong session, from 4-5 pm ET. Speakers are Andy Christensen, the President of Somaden; Jonathan Morris, MD, Neuroradiologist and Director of the Mayo Clinic’s 3D Printing Anatomic Modeling Lab; Beth Ripley, MD, Assistant Professor of Radiology with VA Puget Sound; Justin Ryan, Research Scientist at Rady Children’s Hospital-San Diego; and Formlabs’ Director of Healthcare Gaurav Manchanda.
“The ability to manufacture from the patient’s data (medical imaging or surface scan) has been compelling to a community always looking for ways to innovate. With improving patient care as the primary goal, 3D printing has directly impacted more than 1 million patients. More than 25 years ago, anatomical models began to be used for planning of complex surgical procedures. Today, hospitals are using the technology for surgical guides and more. With increasing numbers of hospitals looking to bring 3D printing into their facility, how are they building the business case?”
Register here.
Additive Industries Hosting Digital Event
On July 9th and 10th, Additive Industries is getting the trade show season running again with what it calls “a corona-proof way to get out of the starting blocks.” At its two-day virtual event, attendees can visit the company’s digital booth, view presentations, and talk to the experts to learn more about the MetalFAB1 3D printer and how the company can help turn your ideas into reality…all without traveling or waiting in line.
“While the virtual domain has limitless possibilities, we still live in the physical world. With our exclusive industry additive manufacturing event – we are making the virtual world a reality.”
Register for the virtual event here.
3D Health Hackathon
The Jersey City Rapid Maker Response Group (JCRMRG), a volunteer collective in New Jersey, is hosting a virtual Community Health Hackathon this week in order to foster community entrepreneurship and take on sustainability, supply chain, and manufacturing challenges that are related to healthcare and PPE (personal protective equipment) during COVID-19. There are three categories: sustainable PPE, modular solution labs, and day-to-day PPE, and the deadline to register is this Friday, July 10, at 12 pm ET. Panelists will meet the nine judges during a Zoom call that night to present their ideas, and then the next two days will be spent hacking. The final submission deadline is July 13th at 9 am, and winners will be announced on July 16th.
“Throughout the COVID-19 health crisis healthcare workers faced critical shortages in PPE created by supply chain disruptions and shortages. Jersey City Rapid Maker Response Group, as well as other groups like them around the country, proved that by quickly deploying 3D-printing capabilities and then extending those capabilities through rapid manufacturing – they were able to scale from producing 1,000 face shields a week to 10,000 face shields a day, both at a fraction of traditional pricing.
“We have reached out to leaders in the tech, manufacturing and 3D-printing communities to form a community-led virtual make-athon. Our collective goal is to continue to bring bright minds together to develop 3D-printing, manufacturing and community-based engineering solutions to address the ongoing needs surrounding supply chain disruptions in emergent and healthcare settings.”
The current prize pool is valued at over $7,500, so what are you waiting for? Register for the hackathon here.
Will you attend any of these events and webinars, or have news to share about future ones? Let us know! Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the comments below.
The post 3D Printing Webinar and Virtual Event Roundup, July 7, 2020 appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
SLA 3D Printing: Formlabs Offers Six New Resins for the Form 3, Form 3B & Form 2
Centered around the miracles of 3D printing, Formlabs tends to have the magic touch—whether individual users or companies are seeking new products like an SLA printer or choosing from a host of high-performance resins. Now, the Somerville, Massachusetts-headquartered manufacturer is releasing a wide range of materials for the Form 3, Form 3B, and Form 2 SLA 3D printers, meant to meet the needs of professionals working in engineering (product design and manufacturing), healthcare, and dental applications.
“Manufacturers have long expressed the desire to replace outsourcing and molding of silicone, urethane, and rubber parts in order to reduce long turnaround times and additional labor,” states Formlabs. “With the launch of our family of Flexible and Elastic Resins, we are expanding our library of high performance, easy-to-use soft materials, and making it possible to produce flexible parts in-house within hours. These resins are well suited for consumer goods prototyping, compliant features for robotics, and special effects props and models.”
Flexible 80A allows for cushioning and absorption and is suitable for engineers 3D printing parts like handles and grips, seals and gaskets—but also for medical professionals developing models displaying cartilage, tendons, and other anatomy like ligaments.
“Balancing softness with strength, Flexible 80A Resin can withstand bending, flexing, and compression, even through repeated cycles,” states the Formlabs team.
Rebranding their original Elastic Resin, Elastic 50A Resin is meant for fabrication of parts like wearables, compressible buttons, enclosures and casings that must stretch, as well as mimicking soft tissue anatomy for 3D models.
“Choose Elastic 50A Resin for parts that will bend, stretch, compress, and hold up to repeated cycles without tearing, and spring back quickly to their original shape,” suggests the Formlabs team.
BioMed Clear Resin is meant for long-term contact with the human body, but can be used for creating jigs and fixtures, as well as medical devices, surgical planning tools, and other R&D tools.
“This USP Class VI certified material is suitable for applications that require wear resistance and low water absorption over time,” states Formlabs.
“Parts printed with BioMed Clear Resin are compatible with common sterilization methods. BioMed Clear Resin is manufactured in our ISO 13485 facility and is supported with an FDA Device Master File.”
Also offering a rigid option for biocompatible 3D prints, Formlabs offers the BioMed Amber Resin. Made in the Formlabs ISO 13485 facility, this new material is suitable for fabrication of medical devices, surgical tools, implants, and other parts and prototypes printed for R&D purposes.
“BioMed Amber Resin is a rigid material ideal for biocompatible applications requiring short-term skin or mucosal membrane contact,” says the Formlabs team.
Dental LT Clear Resin (V2) is meant to be an improvement as Formlabs offers a second-generation biocompatible material for splints and occlusional guards.
“Durable and resistant to fracture, this color-corrected material prints clear, polishes to high optical transparency, and resists discoloration over time for a finished appliance you’ll be proud to deliver,” states the Formlabs team.
Custom Tray Resin is a new material for 3D printing customized impression dental trays, suitable for the following:
- Crowns
- Bridges
- Implants
- Dentures
- Other comprehensive cases
Trays can be 3D printed in under an hour, decreasing time needed for labor in the office or lab with speed in production.
Formlabs is also producing a Temporary CB Resin in four VITA shades, collaborating with restorative dental materials specialists BEGO. This material is meant for direct 3D printing of:
- Temporary crowns
- Bridges
- Inlays
- Onlays
- Veneers
“This durable resin can produce up to 7 unit bridges and temporary restorations for up to 12 months use and enables a digital workflow for collaborative processes between dental technician, dentist, and patient,” explains Formlabs.
See the full availability list for Temporary CB Resin here.
Formlabs also offers a webinar, ‘Deep Dive on Flexible 80A and Elastic 50A with our Materials Product Manager,’ and other helpful information about the new materials at their website. Free samples are available for some materials too.
[Source / Images: Formlabs]
The post SLA 3D Printing: Formlabs Offers Six New Resins for the Form 3, Form 3B & Form 2 appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
New 3D printing jobs at Bluedge, new appointments at Made In Space, Relativity Space, Formlabs, AMT and Poietis
3D Printing News Briefs May 31, 2020: M. Holland, Nexa3D, Formlabs
We’re sharing materials and business news with you in today’s 3D Printing News Briefs. First up, M. Holland is distributing Braskem polypropylene filament, and Nexa3D has launched a new high-performance polymer material, in addition to partnering with 3DZ. Finally, Formlabs is expanding its APAC management teams.
M. Holland Distributing Braskem’s Polypropylene
 Thermoplastic resins distributor M. Holland Company is partnering with Braskem to exclusively distribute the petrochemical company’s new high-performance 3D printing polypropylene (PP) filament, enhancing its own material offerings in order to better support clients with applications development and prototype creation. Polypropylene typically has high failure rates with 3D printing, so M. Holland’s AM engineers tested out Braskem’s PP material first, and found that it generates clean surfaces, great layer adhesion, and better surface performance in support material. It features a proprietary formula, which allows users to achieve low warpage, consistent extrusion, and high stability, in addition to less stringing and more repeatable outcomes.
Thermoplastic resins distributor M. Holland Company is partnering with Braskem to exclusively distribute the petrochemical company’s new high-performance 3D printing polypropylene (PP) filament, enhancing its own material offerings in order to better support clients with applications development and prototype creation. Polypropylene typically has high failure rates with 3D printing, so M. Holland’s AM engineers tested out Braskem’s PP material first, and found that it generates clean surfaces, great layer adhesion, and better surface performance in support material. It features a proprietary formula, which allows users to achieve low warpage, consistent extrusion, and high stability, in addition to less stringing and more repeatable outcomes.
“The 3D printing market has seen increased adoption of additive manufacturing technologies and applications due to machinery commoditization and expanded material offerings. Our partnership to distribute Braskem’s 3D printing polypropylene products will enable injection molders to finally use genuinely 3D printable and easy-to-use polypropylene, opening up a world of applications,” said Haleyanne Freedman, Market Manager, 3D Printing at M. Holland.
Nexa3D Launches High-Performance xCE-Black Polymer
This week, stereolithography 3D printer manufacturer Nexa3D launched its new high-performance polymer material, xCE-Black, which is meant for high-speed printing of injection molding tools, heavy duty parts, and end-use plastic components, such as belt pulleys. Extensive testing by the company found that the single cure polymer exhibits long-term environmental stability and great isotropic properties, which makes it perfect for electronics, industrial, and automotive applications. xCE-BLack also has excellent thermal stability and higher flexural strength.
Brent Zollinger, Customer Success Lead at Nexa3D, stated, “When I needed to produce thousands of belt pulleys in our flexible factory overnight, I selected our new xCE-Black material for the job because of its high flexural strength and long-term stability. Within the first 2.5 hours into the project, I was able to print 2,000 pulleys in a single build or 13 parts every minute. That’s really fast and extremely practical for serial production of industrial parts.”
Nexa3D Announces Reseller Partnership with 3DZ
 More news today from Nexa3D, as the company is entering the southern European 3D printing market thanks to a new reseller partnership with 3DZ Group, the largest AM value-added reseller in that region and an authorized dealer of 3D printers and scanners for some of the industry’s biggest names, including Artec 3D, Formlabs, HP, Markforged, Materialise, and 3D Systems. 3DZ has offered AM services and consultancy to the industry for several years, and will be able to help grow Nexa3D’s global reseller network so the company can increase the distribution of its flagship NXE400 3D printer.
More news today from Nexa3D, as the company is entering the southern European 3D printing market thanks to a new reseller partnership with 3DZ Group, the largest AM value-added reseller in that region and an authorized dealer of 3D printers and scanners for some of the industry’s biggest names, including Artec 3D, Formlabs, HP, Markforged, Materialise, and 3D Systems. 3DZ has offered AM services and consultancy to the industry for several years, and will be able to help grow Nexa3D’s global reseller network so the company can increase the distribution of its flagship NXE400 3D printer.
“It is full speed ahead for us at Nexa3D as we continue to expand our global reseller network, and we’re honored to partner with a company with the vision, reach and proven performance of 3DZ. Manufacturers are adapting processes to become much more agile in their design and resilient in their supply chain in the face of changing circumstances. We believe that our products are at the heart of enabling customers to make that adjustment by converting current 3D printing speeds from dialup internet to broadband-like productivity,” said Avi Reichental, the Executive Chairman and CEO of Nexa3D. “We’re very excited to build together with 3DZ the future of design agility and supply chain resiliency for the benefit of our mutual customers.”
Formlabs Expanding APAC Management Teams
This week, Formlabs announced that it is expanding its management team for the Asia-Pacific (APAC) region by appointing two new hires, in order to continue growing its global operations and scale its growth in these markets. Jiadong Sun, with broad experience leading global sales and marketing at top tech companies across several industries, will be joining the team as the China General Manager, while Yoshinori Hasebe, who brings over 20 years of management experience at multinational companies to the table, will serve as the Japan General Manager. Together, they will help guide the company’s business strategy in these new markets, so that Formlabs users in the APAC region can continue to localize manufacturing and reduce costs and lead times with 3D printing and rapid iteration.
“As Formlabs continues to expand our global operations, Jiadong and Yoshinori will provide strategic guidance as we expand and scale in the important APAC region. These appointments will enable Formlabs to remain at the forefront of 3D printing as needs for additive manufacturing in major industries, including healthcare, continue to grow,” said Luke Winston, Chief Business Officer of Formlabs.
Discuss these stories and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
The post 3D Printing News Briefs May 31, 2020: M. Holland, Nexa3D, Formlabs appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
US Air Force produces on-demand surgical retractor using 3D printing
AMS 2020: Panels on 3D Printing Materials and Applications for Dental Industry
At our recent Additive Manufacturing Strategies 2020 in Boston, co-hosted by SmarTech Analysis, many different topics were discussed in keynotes and panels, such as binder jetting, medical 3D printing, and different materials. Dental 3D printing was also a major topic of discussion at the event, and I attended three panels that focused on additive manufacturing for dental applications.
The first, “Into the dental and oral surgery office,” had three panelists: Dr.-Ing. Roland Mayerhofer, the Product Line Manager for Coherent/OR Laser; CEO Manager of Oral 3D Martina Ferracane; and Mayra Vasques, PhD, a dental prosthesis fellow at the University of São Paulo in Brazil.
Dr. Mayerhofer went first, and provided a quick overview of Coherent’s laser powder bed fusion (L-PBF) systems, and the dental applications for which they can be used.
The versatile CREATOR is the company’s open system, and can print with multiple materials, such as brass, cobalt chromium, steel, and Inconel.
“As long as it works, you can put any powder in you want,” Dr. Mayerhofer said about the 3D printer.
He explained said that the CREATOR setup is “typical but can be as big as a stand-up fridge, not the American double-size.”
You can take a look at the rest of the printer specs above, along with a few features that will be added to the new system that’s coming in 2021, such as two powder hoppers and a build platform.
“Then you can take them out, put fresh hoppers in, and keep going,” Dr. Mayerhofer said.
He stated that the dental field is likely one of the first major adopters of metal additive manufacturing, as the technology offers 100% personalization and can fabricate small, complex parts out of existing materials, like titanium alloys…all perfect features for the dental industry.
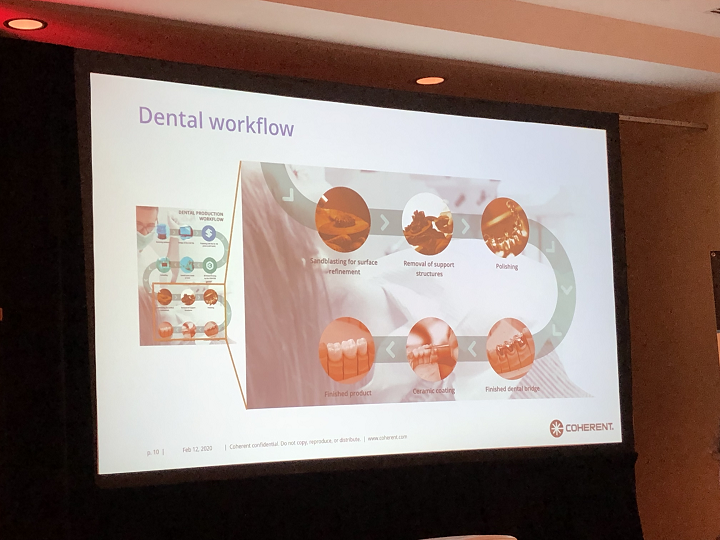
Dr. Mayerhofer then discussed Coherent’s digital dental workflow, which can get from scanning to a completed 3D printed part in 12 steps. Some of these steps include designing the CAD file and preparing it for 3D printing in the company’s APP software suite.
Later process steps are annealing, and then sandblasting, support removal, polishing, ceramic coating – added manually – and voila, you have a finished product.
The Dental Cockpit is Coherent’s latest addition. The CAM software makes it easy to load and print parts, which means that the digital dental workflow as a whole is much less complex. There’s one click to select the file, another to choose the materials and properties, and then a final click to generate the G-code.
Dr. Mayerhofer said that Coherent’s whole dental workflow, 3D printing on the CREATOR include, takes just one work day to fabricate a completed bridge in the dental lab.
After the cast skeleton is scanned, the dental lab begins preparing the CAD data at 8 am. Then the print job has to be prepared in Dental Cockpit, and 3D printing typically begins in the morning.
Once the parts are removed from the print bed, post processing is completed, and then a porcelain coating is added before the product is subjected to heat treatment and polishing. The completed bridge is then ready to go by 4 pm.
Dr. Mayerhofer noted that a dental lab’s ROI on the CREATOR 3D printing system is less than a year…typically about six months, in fact.
Then it was Ferracane’s turn to explain how her company, Oral 3D, makes 3D printing simple for dentists, even as it’s occurring at the industrial level.
“Our solution makes it extremely simple for dentists to bring 3D printing to their practice,” she said.
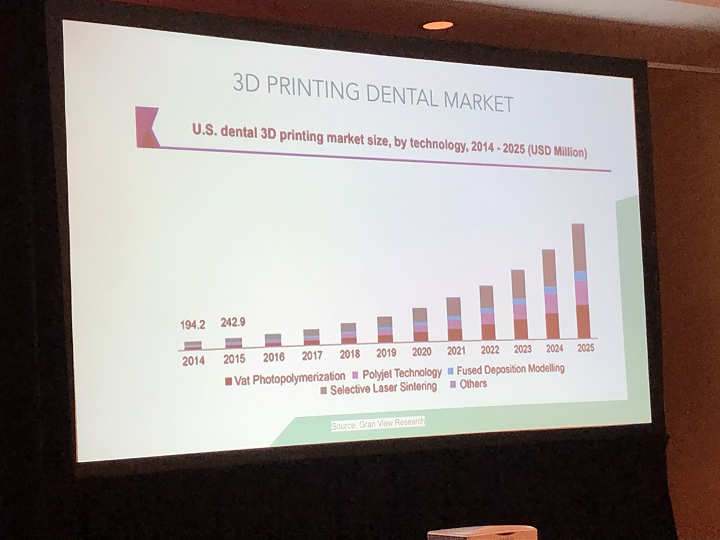
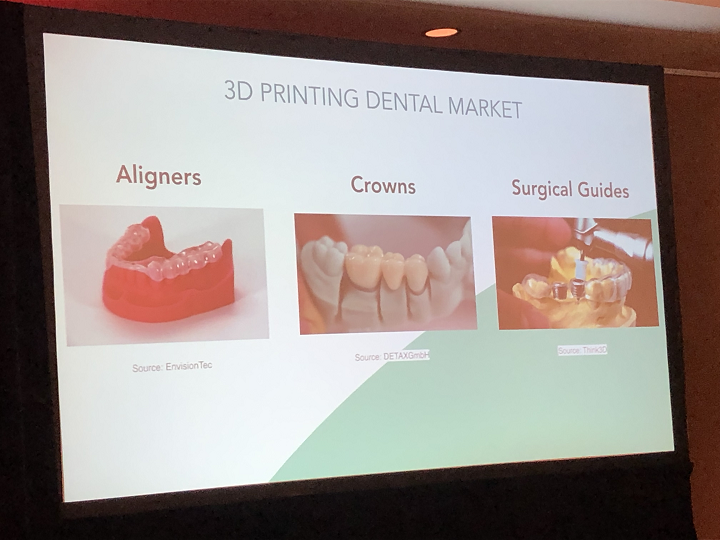
She presented a brief overview of the US dental market, noting that some of the major applications for 3D printing in the field include aligners, crowns, surgical guides, and soft tissue models, which dentists use to test procedures ahead of time.
“Usually today, the way most of these models are done is through intraoral scanning,” she explained.
Ferracane said that SLA technology makes it much easier to make these soft tissue models. But, even so, they can still only be used for testing purposes most of the time.
3D printed models of hard tissue – bone – are also fabricated, but she said that they’re not used often, as it’s difficult for dentists to come up with STL files of just the hard tissue.
She pulled up a slide that had the world “PROBLEM” across the top. The image appears to be scan data of bone, which looks pretty hard to read.
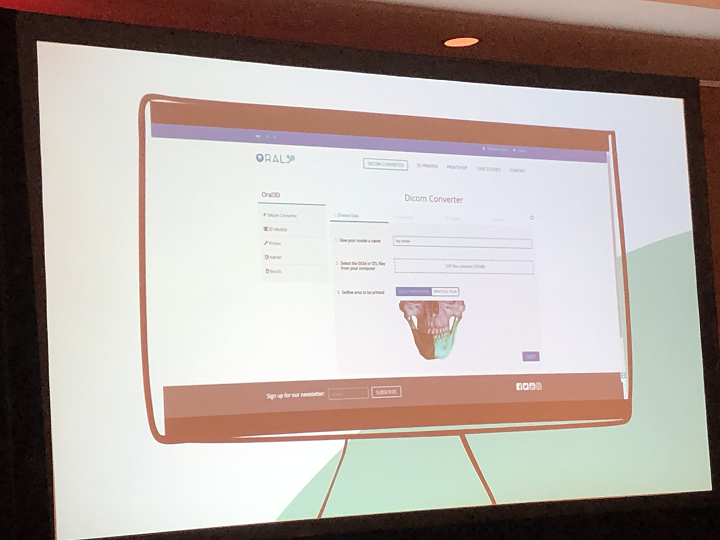
“It’s not easy for dentists to make this into something printable by cleaning up the images,” Ferracane explained. “So they can pay to outsource it to labs to clean it up. But our 3D printing software automatically does this. Just drag the CT scan, and we’ll take care of changing it from DICOM to STL. With one click, we can then convert STL to G-code.”
She said that while it’s obviously good to fabricate dental applications this way using Oral 3D’s printer, it will work with whatever system you’re already using.
These 3D printed models serve a variety of purposes – they can improve communication with patients, help in treatment planning, and even “broaden learning.” Ferracane mentioned that the company has partnerships with NYU and Harvard for this last.
Other applications include bone blocks, made-to-measure titanium membranes, and maxillofacial surgery. Additionally, she stated that Oral 3D recently began collaborating with dental surgeons, who use the company’s 3D printed dental models for planning and patient communication.
She finished by stating that the company believes FDM printing can “be a good value add for dentists.”
Vasques finished things by sharing her research into how things look, dental 3D printing-wise, from the point of view of clinicians.
“It’s common for most to be scared of using 3D printing,” she explained. “They think it’s plug and play, and it’s not.”
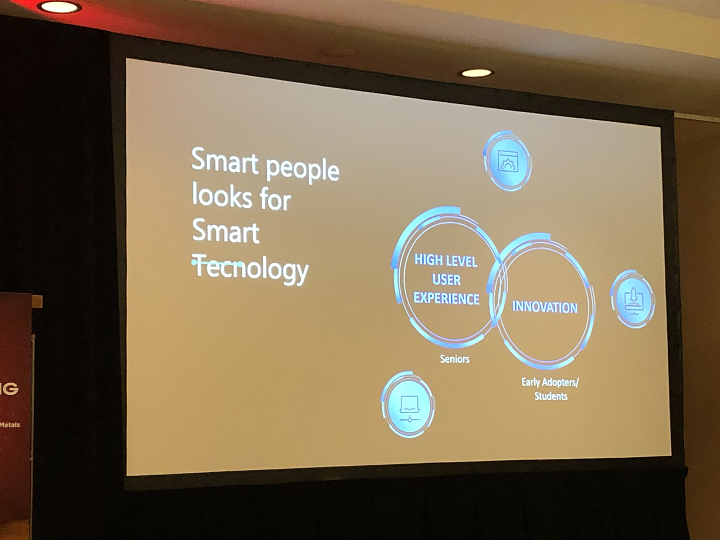
For her research, she divided users into two separate groups – high level experience (seniors), and innovation (early adopters and students).
“We are trying to figure out how these people understand the technology,” she said.
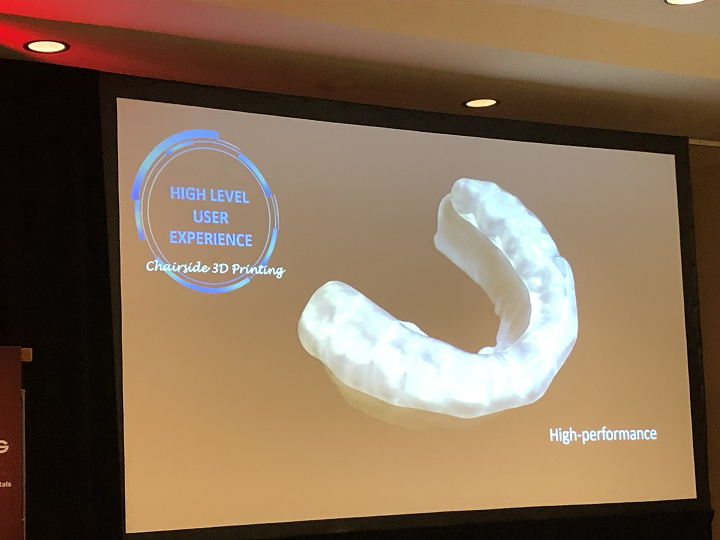
High level users expect accuracy, efficiency, high quality technology, and high-performance materials for the purposes of chairside 3D printing. Vasques said that these users “don’t want to wait 2-3 hours to make products by hand.”
“In university, we’re trying to establish protocols and research to help these people have the results they are expecting.
“We’re trying to solve problems, like mouthguards for sports.”
Vasques said that last year, she and her team published three articles about dental 3D printing topics, such as 3D printed occlusal devices and post-processing. She launched INNOV3D the same year, in order to help train professors in using dental 3D printing.
“We have an online training platform, educational materials, and 3D lab,” she stated.
Once she finished and sat back down, Davide Sher, the panel’s moderator, asked the other two panelists how they would address the challenges that Vasques listed, and how they would make dentists understand more about dental 3D printing.
Ferracane answered that most dentists aren’t buying 3D printers today, because they’re initially taught that the systems are really easy to use when they’re not. Once they run into issues with SLA technology, they get frustrated and just start outsourcing the work instead.
“Then they’re really dissatisfied, because they’re complicated and not just plug and play. We need to help them understand that they can bring the technology back to their office.”
Sher noted that dentists don’t really have the time to learn about the more advanced types, and so asked if the companies directed their technology to users in dental labs; Dr. Mayerhofer said yes.
 After a short break, the next session, “Dental lab experiences with 3D printing,” began. While Les Kalman, an Assistant Professor for Restorative Dentistry at Western University’s Schulich School of Medicine, was unable to make AMS 2020, Arfona founder and CEO Justin Marks and Sam Wainwright, Dental Product Manager for Formlabs, were both ready to go.
After a short break, the next session, “Dental lab experiences with 3D printing,” began. While Les Kalman, an Assistant Professor for Restorative Dentistry at Western University’s Schulich School of Medicine, was unable to make AMS 2020, Arfona founder and CEO Justin Marks and Sam Wainwright, Dental Product Manager for Formlabs, were both ready to go.
Marks went first, explaining that Arfona, founded in 2017 by dental technicians and 3D printing enthusiasts on “the core belief that thermoplastic dental materials should not be substituted for inferior photopolymers,” has been working to “bring 3D printing into the world of dentistry.” The company’s flagship product is its 3D printed flexible nylon dentures.
He pulled up a slide that cited research stating that 36 million Americans are completely edentulous, meaning without teeth, and that 178 million are partially edentulous. But even so, Marks said that there’s an “astronomical” number of people who are still not wearing dentures.
“Most people don’t think about this until it happens to you or someone you know,” he said about missing a tooth. “It’s not always that easy or cheap to fix this with implants.”
According to a survey, only 8% of dentures are digitally fabricated, which means most are still made by hand using analog methods.
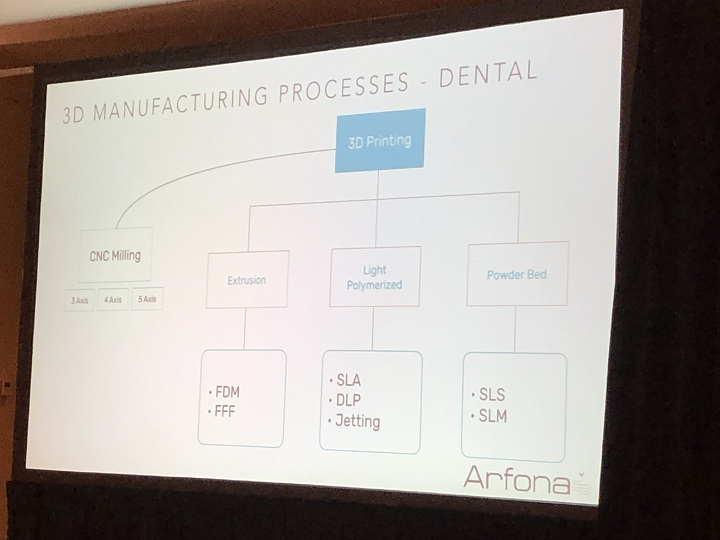
Marks said that even though 3D printing is “becoming more of a buzzword” in the dental industry, most of the materials “have largely stayed the same,” and based on the same technologies and principles. Extrusion-based AM is not used often in dentistry, and powder bed fusion (PBF) is mostly limited to metals, not polymers.
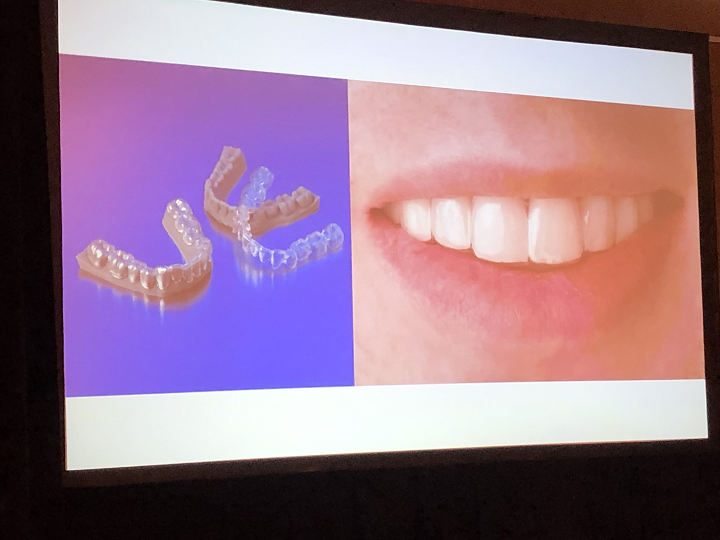
Marks went through a brief history of 3D printing in dentistry. Ubiquitous applications include impression trays, digital models, and resin patterns for casting, while digital dentures are currently happening and things like clear aligners, temporary and long-term crowns and bridges, and multimaterial printing are in development for use in the future.
He said that the ubiquitous ones have one thing in common – they’re used once and then thrown away.
“We’re still not doing much with crowns and bridges,” Marks said. “Clear aligners are the holy grail, and direct printing of the aligner is still a ways off, though all companies are probably working on it.”
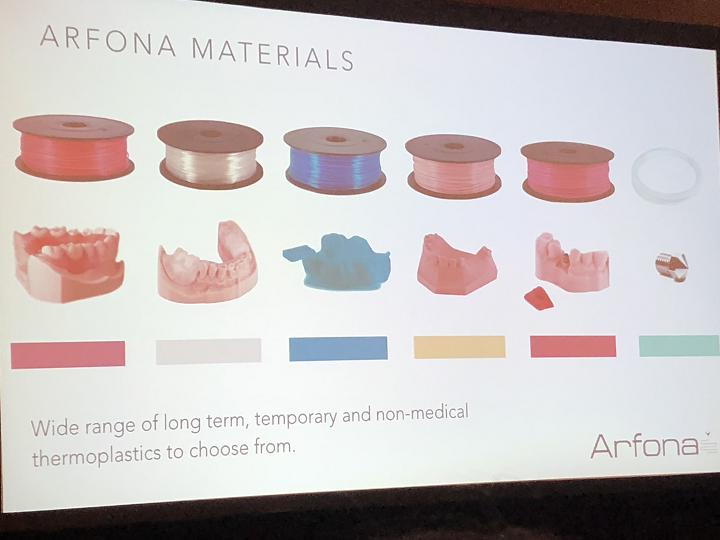
Aronfa’s dental 3D printer is the r.Pod, which is a modified version of a Makerbot clone. The dual extrusion filament system is optimized for all of the company’s thermoplastic materials.
Then it was Wainwright’s turn to talk about dental 3D printing at Formlabs. He agreed with Marks that “FDM and thermoplastics have an incredible place” in the dental industry.
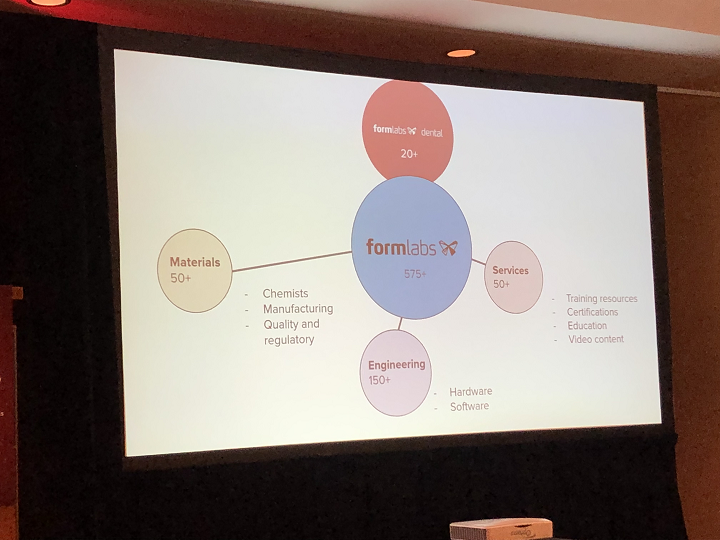
 When the company was founded in 2012, its goal was to make professional-scale 3D printing accessible and affordable for everyone. Now Formlabs employs over 500 people at its multiple locations around the world, and has sold more than 50,000 3D printers.
When the company was founded in 2012, its goal was to make professional-scale 3D printing accessible and affordable for everyone. Now Formlabs employs over 500 people at its multiple locations around the world, and has sold more than 50,000 3D printers.
Wainwright explained that the Form 3B desktop printer, optimized for biocompatible materials, has many dental-specific features, materials, and software, in addition to automated washing and post-curing systems “to help tie in end-to-end dental workflows.”
In addition, Formlabs offers dental materials, and launched its dental service plan (DSP) along with the Form 3B in 2019. Because there are high demands, the 3D printing process is complex, and the DSP offers support.
“We are committed to 3D printing for dental,” Wainwright stated. “We have over 20 people in the dental business unit. But we have the resources of a 500 person-plus company.”
While most are made overseas, Formlabs Dental is now developing photopolymers in my home state, since the company acquired its main material supplier, Ohio-based Spectra Photopolymers, last year. Formlabs’ biocompatible Surgical Guide Resin is the company’s first material made in an ISO-certified facility.
“It’s exciting to have intimate control over design aspects,” Wainwright said.
The image above is an example of the Surgical Guide material. Wainright explained that the light touch supports are very easy to remove, which means that there isn’t a lot of time wasted in post-processing.
He said that 36% of dental labs in the US use 3D printing technology, which makes them very “cutting edge.”
“There’s a ton of market opportunity for dental to go digital,” he said. “We have 30% of this market – we’re the biggest player in dental laboratories and will continue to grow, but compared to Invisalign, it’s not really that much.”
So far, Formlabs has 3D printed more than 10,000,000 parts for the dental industry. Wainwright predicts that in ten years or less, “everything in dental will be 3D printed.”
He reiterated to the room that Formlabs has “a whole host of materials” for dental applications, four of which are solely for fabricating models, which are “really critical to dentists.” As dental offices adopt intraoral scanning technology, it’s helpful to take the scan data and turn it into something physical. Wainwright mentioned that Formlabs’ Grey Resin can achieve fast, accurate prints, and that it’s good for thermoforming as well.
The company’s Draft material is “accurate enough to create models in less than 20 minutes,” which makes it perfect for creating retainers on the same day as a patient’s appointment. Model Resin is good for accurately restoring dental models, while the biocompatible Dental LT Clear Resin can be used to print occlusal splints in addition to models.
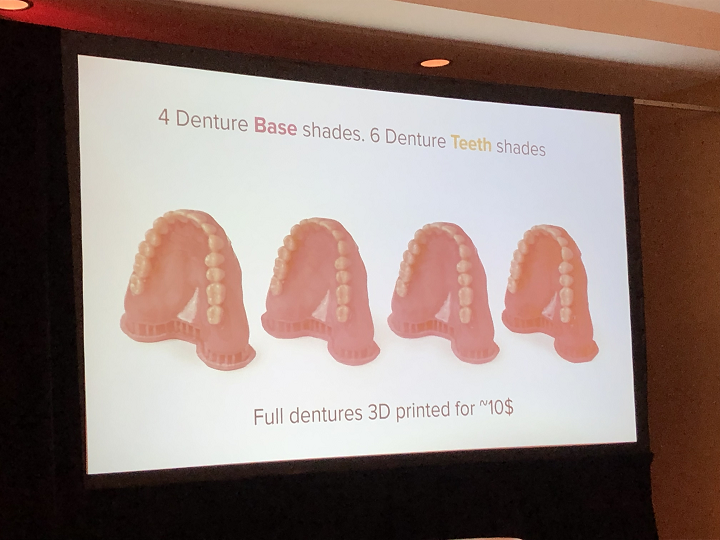
Formlabs’ Digital Dentures solution comes in multiple shades to match a patient’s teeth, and a full set can be 3D printed for less than $10, which Wainwright says is “really a game-changer.”
“We want to make treatments easier, better, and faster,” he said in conclusion.
“3D printing is still very early in dental, this is just the beginning. The materials will just keep getting better, it’s an exciting place to be.”
 Then it was time to eat lunch and chat with other attendees…or, as I did, inhale food and then find a spot in the hallway near an outlet and get a little work done.
Then it was time to eat lunch and chat with other attendees…or, as I did, inhale food and then find a spot in the hallway near an outlet and get a little work done.
After the lunch break, I sat in on my last panel at AMS 2020, “3D materials for dental applications.” It was a panel of one – Gabi Janssen, Business Development Manager and Global Leader, Healthcare Segment Additive Manufacturing, for DSM Additive Manufacturing. She presented on digitalization in healthcare and dentistry.
She tried to play a short movie about what the company does, but due to technical difficulties there was no sound, so she narrated instead, explaining that DSM is “a material company” that also does a lot with nutrition – a brand behind the brands.
The company also has a biomedical department, which helps deliver advanced healing solutions for AM applications, including bioceramics, collagen, polyethylenes, polyurethanes, and hydrophilic coating.
“What we have on the market is filaments,” Janssen said, pulling up a list of the dental materials DSM offers.
Several of the company’s products are geared toward the healthcare market, such as Somos BioClear for dental guides and anatomical models.
“So how do we develop a new material?” Janssen asked. “We’ve discussed 510(k) clearance materials, and you have to work all together. We look at the application, and determine what we need – printer, software, material – to fit what the end user needs.”
She pulled up a slide of the major market drivers in 3D dental printing – performance, mass customization, and time-saving.
“What kind of applications do we have in dentistry?” she asked.
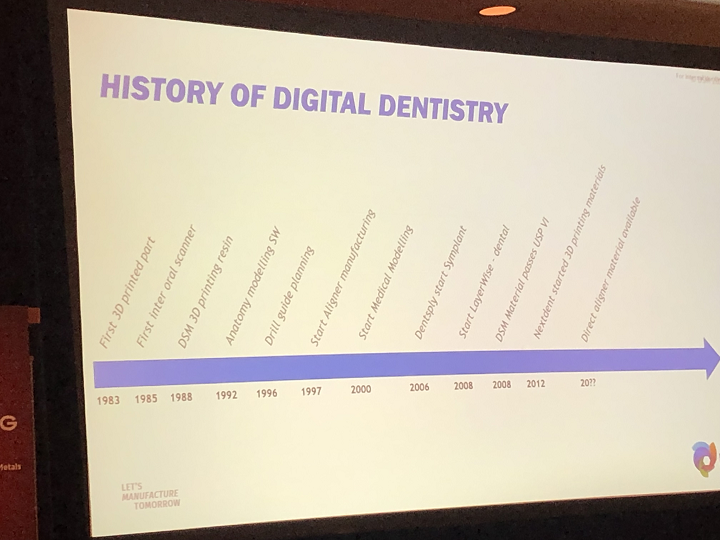
To answer her own question, she showed a brief history of digital dentistry, starting with the first 3D printed part in 1983, moving on to DSM’s 3D printing resin in 1988, the beginning of aligner manufacturing in 1997 and medical modeling in 2000, and DSM’s dental materials passing USP VI in 2008. For 2020 and beyond, hopefully we’ll see the availability of direct aligner materials.
“I think there’s still a lot of data needed to show it’s good,” Janssen said about where the industry currently stands. “Reimbursement is difficult, we need this data to back it up.”
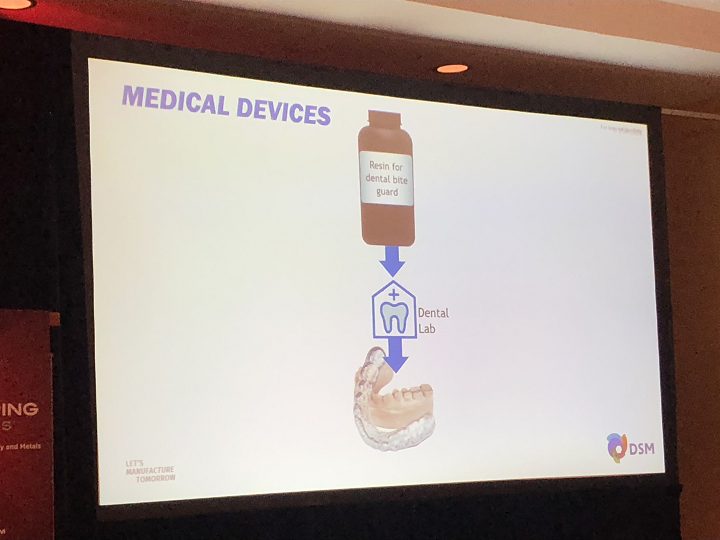
The topic of FDA clearance obviously came up a lot at AMS 2020. Janssen said that DSM has a resin that’s certified for use in dental bite guards, and a general purpose resin that isn’t certified but can be used to make FDA-cleared aligners.
“The end device needs the clearance,” she reminded the room.
She brought up how Materialise was the first company to receive FDA clearance for software about 3D printing anatomical models for diagnostic use. Materialise Mimics inPrint translates the data for the model to the 3D printer. Then, combined with a specific printer and material, it’s possible to fabricate “the model they actually want within a certain safety margin.”
“But, if you want to print medical models, just for patient communication, it does not need to be cleared, because it’s not a medical device,” she explained.
The slide above explains what makes a medical device controlled, i.e. needs clearance, while the below slide lists some very useful definitions, including biocompatibility and risk.
Janssen then brought up the “sometimes confusing standards,” such as ISO standards.
“Depending on what we do with the material, and how long it goes in the mouth, there are different risk associations,” she explained.
In terms of product classification, Class I is the least risky. But, the higher you go up in class, the more research is required to show that the 3D printable material won’t harm patients.
She said that the regulatory industry is changing to have more focus on software, with higher regulations for that software, because it “needs to be validated in combination with the material and equipment.” Additionally, there is more of a focus these days on understanding and managing risks, as well as reducing animal testing…always good news!
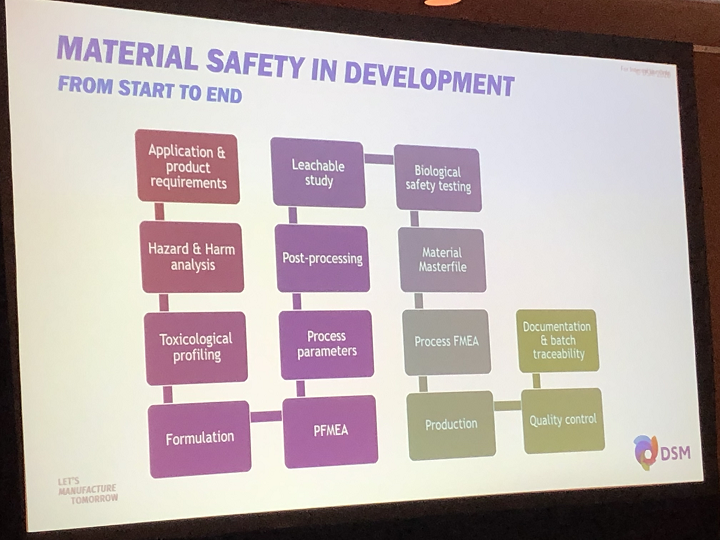
When choosing the proper filaments for your workflow, you should start by working with the dentist on treatment planning. Then, once the patient’s mouth has been scanned, you can create the design in the software. Then the build has to be prepared, which takes some patience and precision – you need to enter the optimal print parameters, and add supports if they’re needed. Then, after the print is complete, it needs to be removed from the bed, supports (if there are any) need to be taken off, and there may even be grinding and painting involved before the final quality check.
“Many process variables can impact the safety of the final end product,” Janssen noted. “So you need to understand the effect the material can have on patients.”
Finally, there are also plenty of steps to follow to ensure material safety in development, so it’s important to follow the instructions your supplier gives you.
Then it was time for some questions. One attendee asked why dentists aren’t all adopting AM, since some products, like mouthguards, look pretty easy to make in the back office.
“This may look easy, but it’s actually not,” Janssen explained.
 She went on to say that the product or device may not always “come out right the first time.” There are a lot of parameters to look at, and potentially tweak, in order to achieve the desired result. A lot of people can get frustrated if it doesn’t work right the first time.
She went on to say that the product or device may not always “come out right the first time.” There are a lot of parameters to look at, and potentially tweak, in order to achieve the desired result. A lot of people can get frustrated if it doesn’t work right the first time.
“What we’re doing now – if you bring your design to us, we’ll do the tweaking for you, as our software has all of the maximum and minimum numbers needed for parameters,” she said.
3D printing thought leader and author John Hornick offered his take on the question, as he has some experience with the matter. He explained that most dental offices are private, though many dentists are consolidating their practices into larger ones, “and their appetite for spending money on these machines may go up.” But, SmarTech doesn’t think the average dentist will spend that much for larger, more expensive 3D printers. That’s why some companies, like Arfona, are working on simpler material extrusion systems.
Another attendee said that it seems like 3D printing companies are just throwing technology at various markets and praying that it sticks. Dentists want to be dentists, and not spend their time dealing with issues like print parameters and melted filament.
“We, as technology providers, need to raise our game and make this work for these people,” Janssen stated.
I think that’s a great note on which to end my AMS 2020 coverage – we, the AM technology providers, need to show the rest of the world how 3D printing can work for their industries.
We hope to see you next winter for Additive Manufacturing Strategies 2021!
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
The post AMS 2020: Panels on 3D Printing Materials and Applications for Dental Industry appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
New Partnership: BEGO’s Dental Materials Allow Formlabs Customers to 3D Print Crowns & Bridges
BEGO, headquartered in Germany, has been a leader in the dental field for 130 years—and as pioneers in 3D printing for the last two decades, they now specialize in all types of dental fabrication techniques for prosthodontics and implant dentistry. Today, they have announced a partnership with Formlabs, the US-based designer and manufacturer of 3D printing systems—along with a wide-ranging product line and other strong partnerships around the world.
 This alliance, just announced, means that Formlabs dental customers will be able to make on-demand dental products for their patients–using BEGO’s dental materials to 3D print both temporary and permanent crowns and bridges.
This alliance, just announced, means that Formlabs dental customers will be able to make on-demand dental products for their patients–using BEGO’s dental materials to 3D print both temporary and permanent crowns and bridges.
 Formlabs customers who are already using the Form 3B and Form 2 3D printers will have access to fabricating permanent single crowns, inlays, onlays and veneers, and temporary crowns and bridges. Appreciating many of the benefits of 3D printing, they will be able to save on the bottom line, while making customized products, faster—and better.
Formlabs customers who are already using the Form 3B and Form 2 3D printers will have access to fabricating permanent single crowns, inlays, onlays and veneers, and temporary crowns and bridges. Appreciating many of the benefits of 3D printing, they will be able to save on the bottom line, while making customized products, faster—and better.
“Directly printing temporary crowns and bridges are one of the most sought-after applications from Formlabs customers. By partnering with BEGO and leveraging their 130 years of dental experience, we will be able to not only address this need but take it a step further by offering materials for permanent crowns. We are excited to see how this partnership can continue to advance the dental industry and overcome the major challenges labs and dentists face as digital dentistry becomes a standard for patient care,” said Dávid Lakatos, Chief Product Officer at Formlabs.
Patients will enjoy:
- Excellent aesthetics
- Less tendency for dental work to age and discolor
- Low-plaque accumulation
- More comfort, thanks to less cold and heat sensitivity
“The ability to use completely digital dental workflows with 3D printed temporary restorations will enable a rapid, low cost, iterative process that improves patient care and case acceptance rates,” states the BEGO team in a press release sent to 3DPrint.com.

Dávid Lakatos, Chief Product Officer at Formlabs with Axel Klarmeyer, Chief Executive Officer of BEGO Dental
The two companies will be in attendance at LMT Lab Day in Chicago from February 21-22, presenting materials for attendees to see and feel, as well as showcasing their new partnership, which is also part of the ‘natural evolution’ of the also recently announced Dental Business Unit and Form 3B, meant to ensure that dentists are able to offer their patients the best in care.
“We could not be happier to partner with Formlabs, especially at this time, where digital dentistry is reaching a breakthrough. It took some time and a lot of effort and commitment of all involved people to be able to offer to the market a fully validated workflow for final restorations. This partnership underlines BEGO’s leading position in the dental 3D printing materials market,” said Axel Klarmeyer, Chief Executive Officer of BEGO Dental.
Find out more about these products here, and please visit BEGO and Formlabs at LMT Lab Day at Formlabs booth (#P-1) and BEGO booth (#H-13).
Both BEGO and Formlabs stay in the news with continue product development and successes from BEGO’s recent launch of the Varseo XL to new resins and other recent acquisitions by Formlabs.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: BEGO]
The post New Partnership: BEGO’s Dental Materials Allow Formlabs Customers to 3D Print Crowns & Bridges appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.