StoneFlower3D launches new Printhead 3.0 for ceramic 3D printing
Third Munich Technology Conference: A reality check on additive manufacturing industrialization
Register for the Third Annual Munich Technology Conference
3D Printing News Sliced: Makerbot, 3D Systems, GE Additive, EOS, ColorFabb, TU Delft, SUTD, AMT
TUM Purchases AMT’s PostPro3DMini for Post-Processing 3D Printed Medical Device Parts
 UK-headquartered Additive Manufacturing Technologies (AMT) is a vertically integrated technology development and manufacturing company that creates automated digital solutions to help its customers unlock the potential of industrial 3D printing. In 2017, when the company was founded, it introduced its PostPro3D commercial offering, which automatically smooths elastomeric and nylon 3D printed parts. The patent-pending technology, which was officially released last year, provides an automated and sustainable post-processing solution for high volume, production 3D printed parts, and works on all types of filament- and powder-based 3D printing methods.
UK-headquartered Additive Manufacturing Technologies (AMT) is a vertically integrated technology development and manufacturing company that creates automated digital solutions to help its customers unlock the potential of industrial 3D printing. In 2017, when the company was founded, it introduced its PostPro3D commercial offering, which automatically smooths elastomeric and nylon 3D printed parts. The patent-pending technology, which was officially released last year, provides an automated and sustainable post-processing solution for high volume, production 3D printed parts, and works on all types of filament- and powder-based 3D printing methods.
Now, AMT has announced the first sale of its new PostPro3DMini system, which was introduced to the market earlier this year. The Institute of Micro Technology and Medical Device Technology (MIMED) of the Technical University of Munich (TUM) confirmed that it has purchased one of AMT’s automated PostPro3DMini post-processing systems, which it plans on using to support its ongoing medical device research.
“We are really pleased to be working with the Mechanical Engineering department at TUM. This is a prestigious research institute that has been working on the progression of AM for many years. The fact that they have purchased the PostPro3DMini to support this research, and for such a demanding application in the medical device sector, is a real testament to the capabilities of the PostPro3D platform and how it can meet the demands for such applications that previously have not been met,” stated Joseph Crabtree, the CEO of AMT.
All of AMT’s post-processing systems are both UL- and CE-certified. The PostPro3DMini is based on the company’s proprietary, automated BLAST (Boundary Layer Automated Smoothing Technology) process, and offers all of the original PostPro3D’s advantages in a more compact unit. It’s a great size for design studios, research institutions, STEM programs, and smaller production runs, and is just as safe and sustainable for polymer 3D printed parts.
Speaking of safety and sustainability, AMT holds these as paramount to its philosophy, and so completed tests on EOS PA2200 3D printed parts processed with its PostPro3DMini. The results conform with all necessary cytotoxicity tests, in addition to skin irritation tests to normative references: ISO 10993-10 (2013), ISO 10993-1 (2018), and OECD TG 439.
The new PostPro3DMini system provides excellent smoothing and surface modification, which is able to achieve a surface quality that’s at least equal to injection molding for 3D printed polymer parts, if not even better. Rather than using water, the process uses a single, recyclable, non-toxic agent instead, and AMT’s automated post-processing hardware is well-suited for applications in medical devices.
 The ISO:13485-certified MIMED at TUM has embraced 3D printing as a viable development and production method for its continued research into new medical devices. That’s why the department was on the lookout for a commercially available system for post-processing when it discovered AMT’s PostPro3DMini.
The ISO:13485-certified MIMED at TUM has embraced 3D printing as a viable development and production method for its continued research into new medical devices. That’s why the department was on the lookout for a commercially available system for post-processing when it discovered AMT’s PostPro3DMini.
MIMED is currently developing individualized instruments for different medical applications using EOS PA2200 material; obviously, as this material is what was tested on the PostPro3DMini, the institute sees a lot of potential for the system. The PostPro3DMini will be integrated into MIMED’s 3D printing process for creating medical devices, in order for the institute to increase its range of SLS medical device parts.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
[Images: Additive Manufacturing Technologies]
The post TUM Purchases AMT’s PostPro3DMini for Post-Processing 3D Printed Medical Device Parts appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.
Solvay Announces Winners of 2019 Solvay AM Cup, First Place Winners Take $10K Home
 It’s that time of year again, as Italy’s Solvay announces winners for what seems to have become a yearly tradition with their AM Cup. For 2019, students were at the ready, and given an industrial task as they were challenged to use Radel® PPSU AM-ready filament for creating an ASTM D638 Type V size tensile bar in the z-axis, along with a wavy-shaped pressure pipe.
It’s that time of year again, as Italy’s Solvay announces winners for what seems to have become a yearly tradition with their AM Cup. For 2019, students were at the ready, and given an industrial task as they were challenged to use Radel® PPSU AM-ready filament for creating an ASTM D638 Type V size tensile bar in the z-axis, along with a wavy-shaped pressure pipe.
While it may seem like an easy challenge to be given an assignment to print out a couple of parts, there was much more to it than that; in fact, students from three continents participated in this contest, with 35 student teams from 32 universities. Solvay’s ultimate goal in initiating the 2019 Solvay AM Cup was to highlight the impact 3D printing materials can have on different applications today due to the high performance of parts—and the availability of different materials and methods. Solvay’s focus was for the students to explore the disruptive technology and learn more about ‘the art of the possible.’
The teams were judged on their collective enterprise in making the parts, judged on:
- Creativity in 3D printing
- Maximum dimensional accuracy
- Mechanical properties
- Performance in burst pressure tests and translucency
Each team was provided with a spool of Radel® polyphenylsulfone (PPSU) AM filament and sent on their way to make plans for winning the competition. Those who were successful in their mission have just been announced:
- First place – The Gekko Performance Team from Technical University (Munich)
- Second place – PPSUsual from Ghent University (Belgium)
- Third place – PPSUPER (Belgium)
“The team secured the first prize due to its ability to achieve 100 percent z-axis strength in the Type V size tensile bar and its wavy pipe showed overall dimensional accuracy, surface uniformity, and a remarkable mechanical performance by enduring a burst pressure test of 1,400 psi (96.5 bar) for two hours,” states Solvay in their press release, also commenting that there was very little separating the teams who won second and third place regarding performance in strength and ductility of their parts.
The winners won $10,000, $5,000, and $3,000, respectively, with the idea that these funds would be well-invested in activities related to higher learning, or ‘societal or entrepreneurial’ endeavors. The 3D printed parts they submitted for the challenge will be on display at the Rapid + TCT show in Detroit, MI (Booth #747) from May 21-23.
“It was inspiring to see the various approaches to solving the challenges of fused filament fabrication (FFF) such as bed adhesion and chamber temperature management. The winning team demonstrated once more that 3D printed parts can virtually match the performance and quality of conventional injection molded parts, provided material, hardware, and process are optimized together,” said Ryan Hammonds, R&D platform manager for Solvay’s Specialty Polymers global business unit and president of the AM Cup Jury.
“We look forward to sharing with our customers the benefits gained from this edition of the Solvay AM Cup for 3D printing the best possible PPSU parts for applications in various industries such as aerospace, healthcare and industrial.”
Along with inspiring students to explore the infinite opportunities available with 3D design and printing, Solvay has continued their momentum, offering strong opinions on the future of 3D printing, expanding materials within their manufacturing processes, and entering into dynamic partnerships. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: Solvay]
Interview: Dr. Christian Haecker, Head of Additive Manufacturing Industrialization, Oerlikon
Newborns with Cleft Lip and Cleft Palates to be Helped Through 3D Printing
 It’s scary to think about newborn babies undergoing surgeries and other medical procedures, but 3D printing has come into play several times to help make these successful. Now, the technology is being used again to help newborns with cleft lip and palates (CLP) in a new study conducted by the Technical University of Munich (TUM).
It’s scary to think about newborn babies undergoing surgeries and other medical procedures, but 3D printing has come into play several times to help make these successful. Now, the technology is being used again to help newborns with cleft lip and palates (CLP) in a new study conducted by the Technical University of Munich (TUM).
According to the study, nasoalveolar molding (NAM) is a “presurgical orthofacial treatment modality” for newborns afflicted with CLP. The method uses a plastic plate to reshape an infant’s gums, nostrils, and lip before undergoing the actual CLP surgery. The plate is inserted and adjusted according to the child’s alveolar development, and then a nasal stent is used to extend the plate during treatment. The technique is considered complete once the primary surgical closure of the lip is performed.
While NAM does work, the treatment is lengthy, with many weekly adjustments; it also requires that several impressions be taken when the baby needs new plates, and the treatment modality is only offered in special centers. However, by introducing CAD/CAM technology and 3D printing into the NAM treatment, the amount of impression-taking, along with the cost of the treatment, can be decreased, and the production modality is better facilitated.
A team of researchers based at TUM recently published a paper, titled “A semi-automated virtual workflow solution for the design and production of intraoral molding plates using additive manufacturing: the first clinical results of a pilot-study,” in the Nature journal.
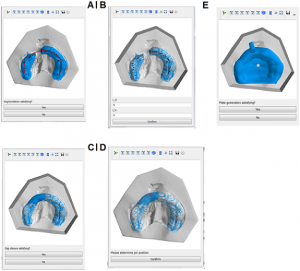
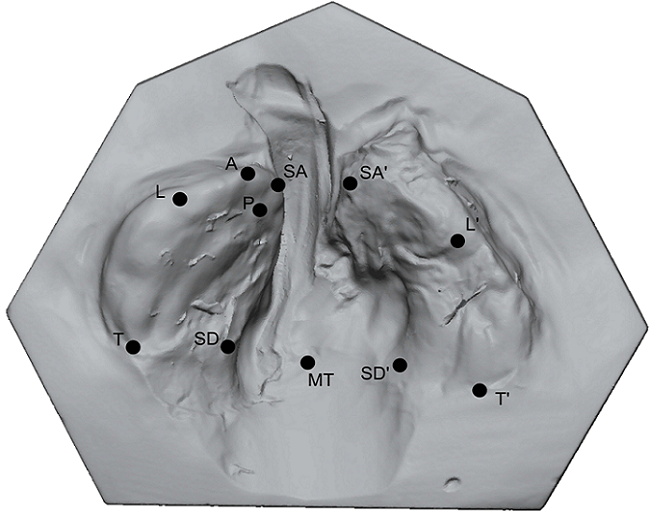
Graphical User Interface for the design of RapidNAM devices. (A) Automated detection of alveolar crest. (B) Selection of bridging area. (C) Gap closure. (D) Pin positioning. (E) Virtual plate.
The abstract reads, “Computer-aided design and computer-aided manufacturing (CAD/CAM) technology has been implemented in the treatment of cleft lip and palates (CLP) by several research groups. This pilot study presents a technique that combines intraoral molding with a semi-automated plate generation and 3D-printing. The clinical results of two intraoral molding approaches are compared. This is the first clinical investigation of semi-automated intraoral molding. Our study included newborns with unilateral CLP. Plaster models were digitalized and measured by two independent observers. Two methods of CAD/CAM-assisted intraoral molding were compared: (i) stepwise manual design of molding plates (conventional CAD/CAM-intraoral molding) and (ii) a semi-automated approach with an automated detection of alveolar ridges (called RapidNAM) assisted by a graphical user interface (GUI). Both approaches significantly narrowed the clefts and resulted in a harmonic alveolar crest alignment. The GUI was easy to use and generated intraoral molding devices within minutes. The presented design solution is an efficient technical refinement with good clinical results. The semi-automated plate generation with a feasible GUI is fast but allows individual adaptations. This promising technique might facilitate and foster the more widespread use of CAD/CAM-technology in intraoral molding therapy.”
In their study, the researchers described their virtual workflow, and also analyzed how effective semi-automated intraoral molding plate generation, or RapidNAM, is for helping to treat CLP.
“Healthy newborns with unilateral CLP (n = 14) were included in the study,” the researchers wrote in their paper. “Two groups were formed: one group was treated with conventional CAD/CAM-intraoral molding plates as published previously with digitally designed intraoral molding plates serving as a reference group and the other group with RapidNAM-plates. In both groups, impressions were taken from the upper jaw within the first few days of life and at the end of molding therapy when primary lip closure was performed at the age of approximately 3–4 months.”
A 3D triangulation scanner from 3Shape in Denmark was used to digitalize the casts, and after creating a graphical user interface (GUI), an algorithm automatically detected the alveolar ridge, in order to find the monthly growth rate in the anatomical study of 32 healthy newborn babies. Special 3D software was used to help with plate expansions during the manual plate molding.
The study concludes, “RapidNAM overcomes previous limitations of conventional CAD/CAM-intraoral molding plates by its semi-automated workflow. The GUI creates a series of molding plates within a few minutes but still allows changes by the user. The resulting plates are as adaptable as conventional NAM-devices. The algorithm automatically detects the edentulous alveolar ridges and may also have further dental applications. RapidNAM gives good clinical results and may bring nasoalveolar molding to a broader practice.”
Co-authors are Florian D. Grill, Lucas M. Ritschl, Franz X. Bauer, Andrea Rau with the Friedrich Alexander Universität Erlangen-Nürnberg, Dominik Gau, Maximilian Roth, Markus Eblenkamp, Klaus-Dietrich Wolff, and Denys J. Loeffelbein.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.