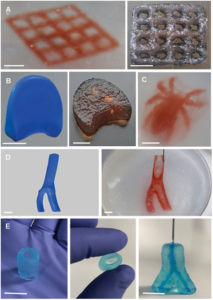
Researchers continue to experiment with materials and extrusion techniques, introducing the use of chaotic flows for improved microarchitectures in the recent study, ‘‘Using chaotic advection for facile high-throughput fabrication of ordered multilayer micro- and nanostructures: continuous chaotic printing.’
The authors present a unique form of fabrication with a method that allows for more precise control of the structure and surface area being printed through the use of what they call ‘continuous chaotic printing.’ The process relies on a combination of chaos theory and fluid dynamics, in which the flow of material develops into complex fractals. To achieve this, the team created chaotic material flows by reorienting and splitting fluid as it is mixed, as shown in the figure below.
With a focus on multiple materials, multiple layers, and fabrication of varying cells in proximity to each other on larger surfaces, their goal was to test the potential for chaotic printing in a variety of applications, but with a special focus on bioprinting and biomedical research. Building on previous research with chaotic flow, the authors experimented with a chaotic printer (paired with a Kenics static mixer), fabricating fibers made from alginate.
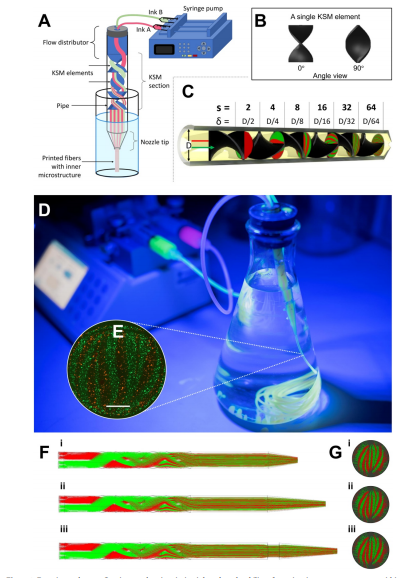
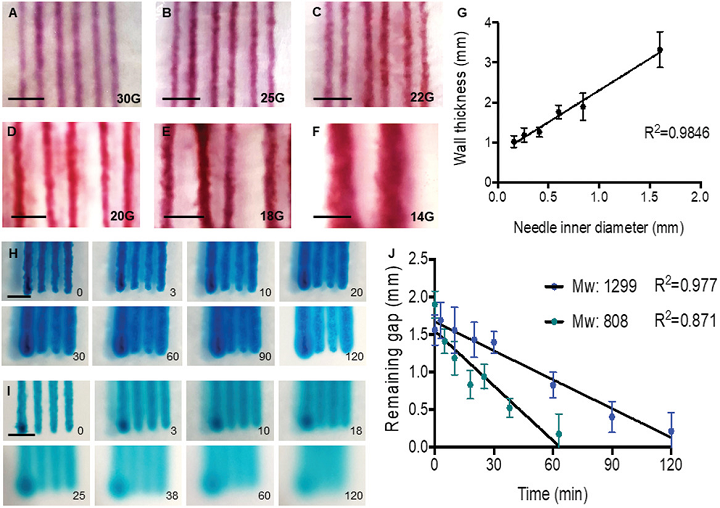
Experimental setup. Continuous chaotic printing is based on the ability of a static mixer to create structure within a fluid. The Kenics static mixer (KSM) induces a chaotic flow by a repeated process of reorientation and splitting of fluid as it passes through the mixing elements. (A) Schematic representation of a KSM with two inlets on the lid. The inks are fed at a constant rate through the inlets using syringe pumps. The inks flow across the static mixer to produce a lamellar structure at the outlet. The inks are crosslinked at the exit of the KSM to stabilize the structure. Our KSM design includes a cap with 2 inlet ports, a straight non-mixing section that keeps the ink injections independent, a mixing section containing one or more mixing elements, and a nozzle tip. The lid can be adapted to inject several inks simultaneously. (B) Two rotated views at 0◦ and 90◦, of a single KSM element. (C) 3D design of a KSM with 6 elements and schematic representation of the flow splitting action, the increase in the number of striations, and the reduction in length scales, in a KSM-printhead. The resolution, namely the number of lamellae and the distance between them (δ), can be tuned using different numbers of KSM elements. (D) Actual continuous chaotic printing in operation. The inset (E) shows the inner lamellar structure formed at the cross-section of the printed fiber (the use of 4 KSM elements originates 16 striations). Scale bar: 250 µm. (F) Longitudinal or (G) cross-sectional microstructure of fiber obtained using different tip nozzle geometries. Images show CFD results of particle tracking experiments where two different inks containing red or green particles are coextruded through a printhead containing 4 KSM elements. The lamellar structure is preserved when the outlet diameter is reduced, from 4 mm (inner diameter of the pipe section) to 2 mm (inner diameter of the tip), through tips differing in their reduction slope.
An unlimited number of inks can be used in chaotic printing, but for this study, the researchers used basic techniques, with sodium alginate as a base. Experiments were then performed with composite inks featuring suspensions like polymer microparticles, graphite microparticles, mammalian cells, or bacteria. Ultimately, the team reported they were able to print ‘fine and well-aligned microstructures’ at high extrusion speeds of 1–5 m of fiber/min.
“This printing strategy is also robust across a wide range of operation settings. We conducted a series of printing experiments at different inlet flow rates to assess the stability of the printing process. As long as the flow regime is laminar and the fluid behaves in a Newtonian manner, the quality of the printing process is not affected by the flow rate used in a wide range of flow conditions,” explained the authors.
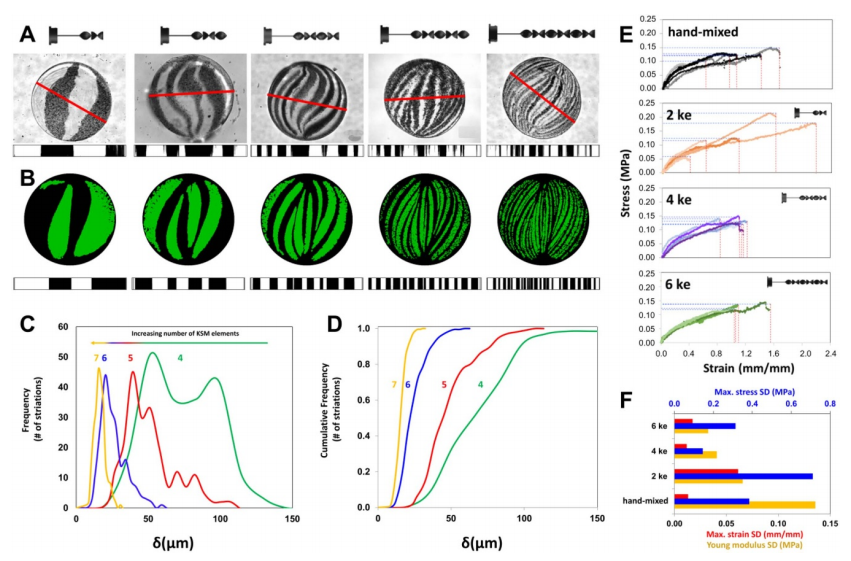
Evaluation of the striation profiles and mechanical properties of chaotically printed alginate/graphite fibers. (A) Lamellar microstructure of fibers produced with printheads containing 2, 3, 4, 5, or 6 KSM elements. The thickness of each lamella, along the red line, was determined by image analysis using Image J (shown below each cross-sectional cut). Scale bar (red): 2 mm. (B) The microstructure at each cross-section was reproduced by CFD simulations, and the thickness and position of each lamella was calculated. (C) Striation Thickness Distribution (STD) and (D) cumulative STD for constructs printed using 4, 5, 6, and 7 KSM elements. (E) Comparison of stress-strain curves of fibers fabricated by extrusion of pristine alginate and graphite without chaotic mixing (marked as hand-mixed) or with chaotic printing using 2, 4, or 6 KSM elements (marked as 2, 4, or 6 ke). (F) Comparison of the standard deviation of tensile properties (i.e. maximum stress, maximum strain, and Young’s modulus for the same set of fibers; 5 fibers per treatment).
The researchers also noted the following while printing:
- Stable fibers were printed using a cone-shaped nozzle tip with an outlet diameter of 1 mm, with flow rates ranging from 0.003 to 5.0 ml min−1.
- Printheads with varying geometries did not affect structures being printed.
- Computational fluid dynamics (CFD) showed that the inclination of the nozzle tip also did not affect the materials.
Continuing to emphasize the ‘robust’ qualities of chaotic printing—especially when used with small nozzles—the researchers were able to control resolution, noting also that, due to the deterministic qualities of chaotic flows, fabrication of structures was ‘fully predictable.’ Resolution was in part illustrated by the number of gill-like ridges, or ‘lamellae,’ created through the chaotic mixing process.
“As the number of elements used to print increased, the number of lamellae observed in any given cross-sectional plane of the fiber also increased, while the thickness of each lamella decreased,” explained the researchers. “Therefore, users of continuous chaotic printing will have more degrees of freedom to determine the multi-scale resolution of a construct, as this is no longer mainly restricted by the diameter of the nozzle (or the smallest length-scale of the nozzle at cross-section).”
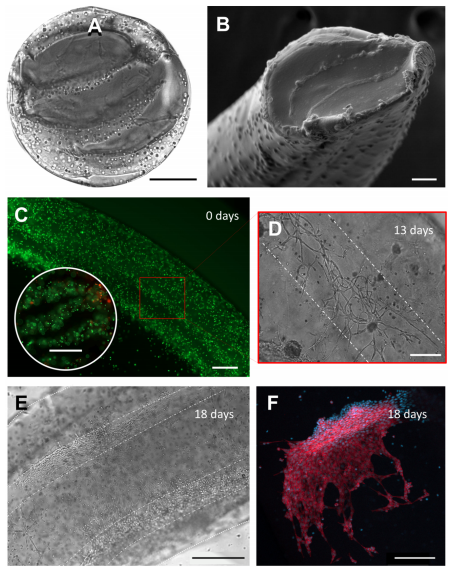
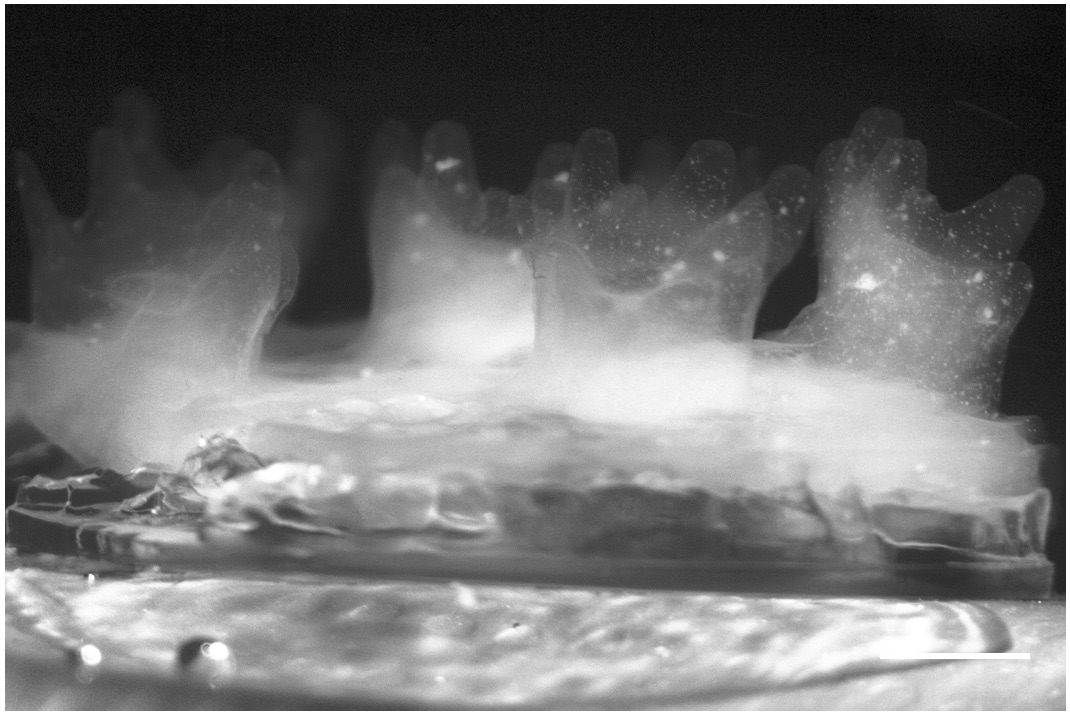
Bioprinting of living micro-tissues: (A) Optical and (B) SEM micrographs of the cross-sectional view of a construct in which C2C12 cells are chaotically bioprinted in an alginate/GelMA hydrogel using a 3-KSM printhead; Scale bars: 500 µm and 50 µm, respectively. (C) Longitudinal view of a chaotically bioprinted construct; a high cell viability is observed at the initial time, as revealed by a live/dead staining and fluorescence microscopy. Scale bar: 500 µm. Inset shows a cross-sectional cut. Scale bar: 500 µm. (D) Cells spread along the chaotically printed striations, preserving their original positions after 13 d of culture. Scale bar: 200 µm. (E) Optical microscopy view of a segment of fiber containing C2C12 cells 18 d after printing. Scale bar: 500 µm. (F) Close-up of a region stained to reveal F-actin/nuclei, showing the cell spreading and the formation of interacting cell clusters. Cell nuclei can be identified as blue dots. Actin filaments appear in red. Scale bar: 200 µm.
Further, as the researchers began experimenting with bioprinting, they were able to create alginate fibers rich with cells, and lightly enriched with protein to encourage ongoing sustainability. This is one of the greatest challenges in tissue engineering. Even though the correct materials, techniques, and concepts may be in place, if researchers cannot keep the cells alive long enough, they must go back to the drawing board.
Altogether, the team suggests that it has developed a completely novel way to control the resolution of an extrusion-based printing system. As shown in the image below, not only can this continuous chaotic printing process control print resolution through the diameter of the nozzle, but at the microscale as well through the chaotic mixing techniques deployed.
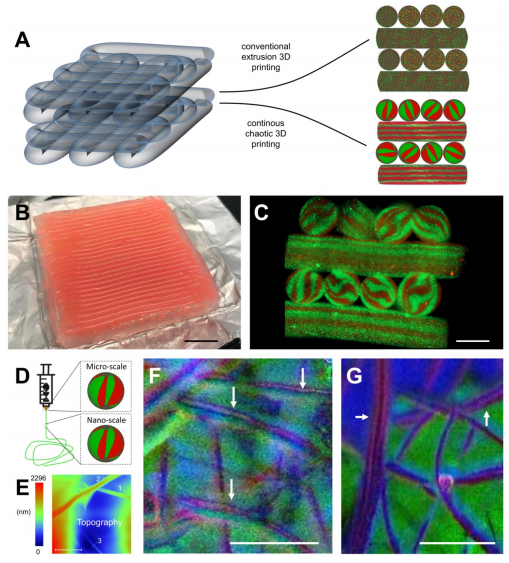
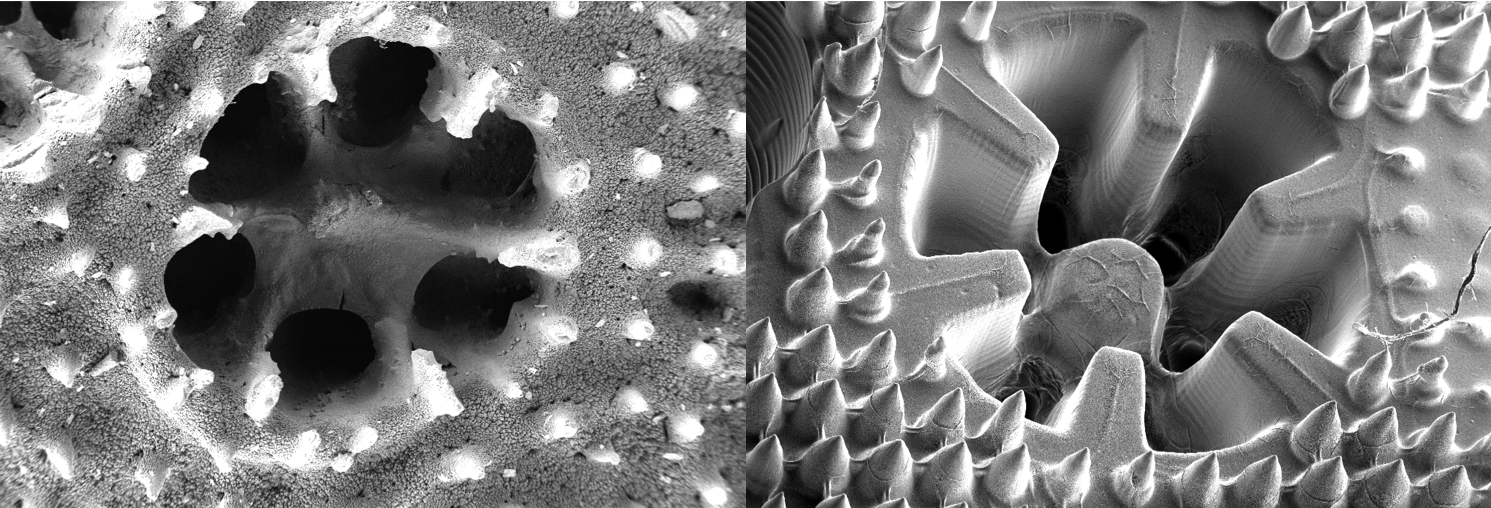
Development of multi-scale architectures based on 3D continuous chaotic printing: (A)–(C) 3D printing of hydrogel constructs using a KSM-printhead integrated to a commercial cartesian 3D printer. (A) Schematic comparison of the lack (prepared using conventional extrusion techniques) and presence of internal lamellar microstructures (developed using continuous chaotic printing). (B) Printing of a long fiber arranged into a macro-scale hydrogel construct (3 cm × 3 cm × 4 mm). Scale bar: 5 mm. (C) Transverse cut of the macro-construct showing the internal microstructures. Scale bar: 1 mm. (D)–(G) Chaotic printing of fibers coupled with electrospinning. (D) Schematic representation of the coupling between continuous chaotic printing and an electrospinning platform; an ink composed of a pristine alginate ink (4% sodium alginate in water) and an ink composed of a polyethylene oxide blend (7% polyethylene oxide in water), were coextruded through a chaotic printhead and electrospun into a nanomesh. (E) AFM image showing the diameter of three individual nanofibers ((1) 0.82 µm, (2) 1.05 µm, and (3) 0.437 µm) within the electrospun mesh. Scale bar: 5 µm. (F), (G) photo-induced force microscopy (PiFM) reveals the lamellar nature of the nanostructure within a nanofiber (white arrows) originated using (F) a 2-element KSM printhead, and (G) a 3-element KSM printhead. Scale bar: 1 µm.
The researchers noted that not only could this have a profound impact on multimaterial printing technologies, but on bioprinting as well:
“Our results demonstrate the unrivaled ability of chaotic printing to deploy cells within high SAV fibers. As available bioprinting and bioassembly technologies approach the resolution and SAV of chaotic printing, they also tend to require long fabrication times and mechatronically coordinated control systems,” concluded the researchers. “In addition to multicellular, high SAV constructs, chaotic printing offers other breakthroughs in regards to currently available multimaterial printing technologies that, typically, require optimized inks that must be deployed under a specific and narrow range of conditions.”
[Source / Images: ‘Using chaotic advection for facile high-throughput fabrication of ordered multilayer micro- and nanostructures: continuous chaotic printing’]
The post Continuous Chaotic 3D Printing: Using Chaos Theory to Control Microstructures appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.