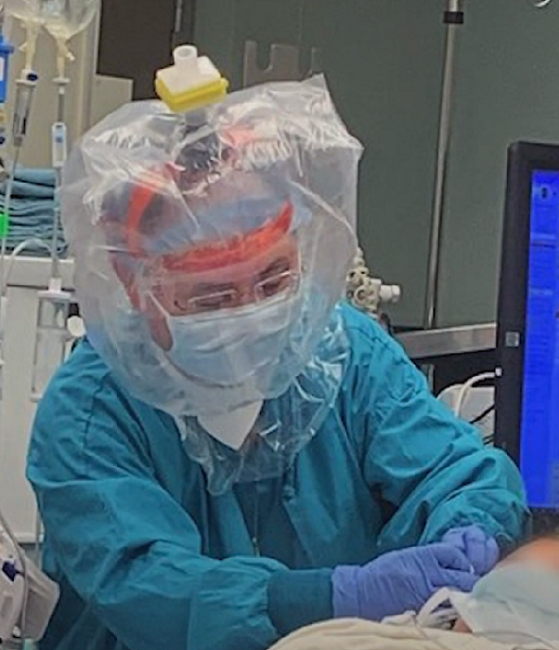
Due to the ongoing COVID-19 crisis, this year’s 3DEXPERIENCE Forum by Dassault Systèmes had to be re-imagined as a virtual event, just like so many other conferences. At 1 pm EDT on July 29th, nearly two months after the in-person event was meant to have taken place in Florida, the company began the live stream of the Plenary Session for “3DEXPERIENCE: A Virtual Journey,” a series of digital programming that replaced the annual North America customer event.

Unfortunately, the webinar seemed to be having issues, which continued on and off over the next two hours of the live stream, so I missed pieces here and there. Technical difficulties happen all the time at live events, too, so the only real difference here was that I couldn’t raise my hand and say, “I’m sorry, the audio and picture cut out, could you repeat that please?” Luckily, Dassault had the webinar up to view on-demand the very next day, so I was able to go back and check out the parts that I had missed.
Erik Swedberg, Managing Director, North America, Dassault Systèmes, got things started with his segment on “Business in the Age of Experience: Challenges and Opportunities for North America,” which focused on manufacturing and supply chains, and why companies looking to transform, some sooner than they’d hoped due to the pandemic, should “invent the industry of tomorrow,” rather than trying to digitize the past or the present.

“Yesterday, businesses focused on automation of the manufacturing system; this is Industry 4.0. Today, many industrials are digitizing the enterprise system. It’s not enough. You need to create experiences. Tomorrow, the game changers will be those with the best developed knowledge and know-how assets. Why? Simple. Because the Industry Renaissance is about new categories of new industrials creating new categories of solutions for new categories of consumers,” Swedberg said.
He mentioned Tesla and Amazon, companies in Silicon Valley working to create autonomous vehicles, and fab labs creating and printing smart, connected objects.
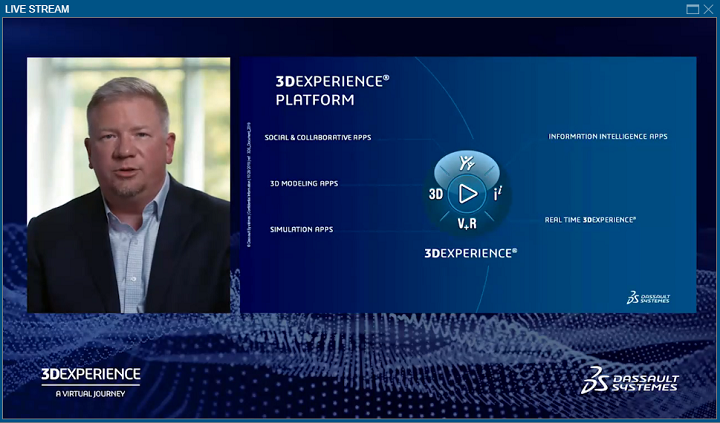
“The 3DEXPERIENCE platform is a platform for knowledge and know-how—a game changer, collaborative environment that empowers businesses and people to innovate in an entirely new way,” he continued. “Digital experience platforms for industry, urban development, and healthcare will become the infrastructure for the 21st century.”
Swedberg explained how 3DEXPERIENCE can allow any business to become social, by connecting employee innovation into the system where the company’s products are designed. This was a common theme today, which you’ll be able to see later.
He also explained that, with Dassault’s 13 brand applications—such as SIMULIA, CATIA, and SOLIDWORKS—the company can serve a wide variety of industries, helping its customers on their journey to invent tomorrow’s industry.
“In summation, we are in the experience economy, the Industry Renaissance is here, and world events are accelerating the need for digital transformation. As the world changes, we will partner with you for success,” Swedberg concluded. “We have the people and the insights to help you on your journey.”

Dassault’s Vice Chairman & CEO Bernard Charlès was up next, speaking about “From Things to Life.” He first said that he hoped no one on the live stream, or their loved ones and colleagues, had been impacted by the COVID-19 crisis.
“We’ve gone through a tough time, all of us. And we are with you, and we are learning a lot also from the crisis,” Charlès said.
Even though I’ve worked from home for nearly four years now, other aspects of my life have been turned upside down in the last few months, and I felt a kind of solidarity whenever the session’s speakers brought up how all of our lives, and our industry, have changed. Charlès also congratulated everyone signed into the live stream on working together, and continuing to innovate, during the pandemic; the continuing health crisis was another theme that threaded throughout the plenary session.
He said that the 3DEXPERIENCE platform is about inclusiveness, “because it means ideas and people connecting.” He shared some of the work that 3DEXPERIENCE users had accomplished during the recent and varied quarantines, such as creating respirators, improving logistics, and working to make the quality of airflow in hospitals better. He said that all of these projects were done on the 3DEXPERIENCE cloud.
“So many of you accelerated the cloud implementation, to be able to work from anywhere, especially from home, during confinement time.”
He mentioned that we are moving from a product economy to an experience economy, and that, in the long run, companies will continue to produce, and maintain ownership of, products and services throughout the life cycle, while their customers will get to enjoy the experience.
“That will accelerate innovation for a sustainable world,” Charlès said.
Next, he talked about a few companies that have been using the 3DEXPERIENCE platform for interesting projects, like California-based Canoo, which dreams about refining urban mobility with an electric vehicle that can be used as a service or subscription, rather than being owned by individuals.

In order to create innovation, Charlès said, you need to be sure that your digital platform will work, and Canoo stated that 3DEXPERIENCE hit the mark here, helping to speed things up in the product development process.
He then talked about Arup, a company that’s using the 3DEXPERIENCE platform to create a virtual Hong Kong for city planning purposes. Arup is working to make Hong Kong a smart city, and the platform is helping the company in this endeavor; for example, Arup and Dassault just completed a project called the Common Spatial Data Infrastructure Built Environment Application platform…say that three times fast.
Finally, Charlès explained that the role of life sciences is to “protect what we care about,” and said that industry pioneers are coming up with new and different ways to diagnose and care for people. He stated that creating new healthcare experiences is a complex project, because it means converting big data into smart data and simulating real world situations in a virtual world. Luckily, 3DEXPERIENCE can help with this.
“3DEXPERIENCE…is a system of operation, because the platform can help you run your business, and the platform should also help you invent a new business model,” Charlès concluded. “The common values across all the industries we serve is putting the human at the center of everything we do.”
Next, Renee Pasman, Director of Integrated Systems at Skunk Works for Lockheed Martin, provided an overview of using the digital thread, and the 3DEXPERIENCE platform, for the product lifecycle, “and how Lockheed Martin is leveraging it to drive increased affordability, efficiency and collaboration throughout the lifecycle.”
“…Our projects cover the entire product life cycle that you might imagine from an aerospace and defense type of program, all the way from conceptual design through modeling and simulation, manufacturing, to sustainment and end of life,” she explained. “And one key part of the Skunk Works culture in the last 75 years has been very close collaboration across all of those areas. What we’ve learned as we have started this digital thread initiative is that by giving our workforce these latest tools, we’ve been able to make that collaboration easier, to be able to make it go faster, to be able to bring data in sooner, make better decisions, see what the impacts are of those decisions, and use that to guide where we are going.”

She explained that the product lifecycle “really starts with design,” and said that by starting this new Near Term Digital Thread/Affordability initiative and giving its workforce the 3DEXPERIENCE tools, Skunk Works has learned that collaboration is faster and stronger, and that we “make better decisions to guide where we’re going.”

We’ve all heard about this issue before—there are two versions of an important product document, and some people update one, while others update the other, and no one has a clear idea of which version is correct and most up-to-date. It’s frustrating to say the least. But Pasman noted that by using the 3DEXPERIENCE product lifecycle management platform, “we’re starting to see efficiency benefits now.”

Pasman also said that the Skunk Works team has learned something “unexpected” with the platform, and that’s the social collaboration it provides, which allows users to “make changes with a level of certainty.”
“We hadn’t necessarily focused on this area, but our teams really used this environment to collaborate better, and found it to be very useful to have all information in that single source of truth.”

Pasman also noted the usefulness of having a life cycle digital twin, as it “allows us to tie it all the way back not just to manufacturing but actually back into design, and making sure the data flows in the digital twin seamlessly.”
“I think if you talk to maintainers or sustainment and users, there’s a lot of time spent putting data into different systems. By making it easier to do that, it allows people to focus on the hard parts of their job, and not just the data entry parts,” she explained. “Collaboration between different areas and getting data flowing is where we see a lot of the benefit from 3DEXPERIENCE, from affordability and product quality perspectives. We’re focused now on how to take the next step in this journey and improve schedule and affordability to fit into the market space that we are working in today. That’s where a lot of the work from our digital thread initiatives have been focused.”

Next up, Craig Maxwell, the Vice President and Chief Technology and Innovation Officer for Ohio-based motion and control technologies leader Parker Hannifin, spoke about “Simple By Design.” The multinational company has been integrating some of the tools that Dassault has been developing over the past few years, which has been valuable to the company.
“When we look at any enterprise or business, we saw these as opportunities that would manifest themselves as complexity,” he said in reference to the image below. “An average customer experience, which might be the ability to ship on time, with high and consistent quality. Of course, inconsistent delivery would manifest itself as complexity. High cost would be complexity…and then all of this would beget complexity in its many forms.”

GIPI = Global Industrial Performance Index
He said that all of these complexities can add up to new opportunities to take the company on the path to high performance. Maxwell also explained that the company’s traditional simplification efforts had revolved around design and organizational structure, explaining that 80% of any business’s profits and sales come from 20% of its portfolio.

“So by slicing and dicing that, could we eliminate complexity? The answer is a resounding yes,” Maxwell said.
He explained that 70% of a product’s cost is design, while 30% is labor and overhead, like lean manufacturing and the supply chain. The key is to spend less time on L&O, or conventional simplification, and work harder to reduce business complexity in that 70% design range. He said there are hundreds and thousands of decisions made on the L&O side, which, while easier to change, had a more limited impact on the long life cycles of their products.
“There were processes in place that we felt could address that reactively, not proactively,” he said.
With design, the decisions made were “relatively few and quick,” even though they could make a significant impact, because they would be difficult to change, mainly due to expensive tooling.
“We believe that if we can address design complexity, it would enable us to move faster and to grow by taking market share,” Maxwell said.

He explained that the cross-functional team Parker Hannifin set up to address “new” product complexity in a proactive way knew early on that there are two different value streams of Simple by Design.
“New products, for sure, but also core products,” he said. “If you look at where the money is, new products get a lot of attention, but our business is core products…they’re undergoing revisions constantly because our customers are asking for things that are different.”
The team decided to tackle new products first, and spent a lot of time working on design-related objectives, which is where they thought “a lot of the complexity and cost was being created.” He explained that the team wanted to keep the customer at the center of their attention, figure out what their pain points were and what they wanted, and get rid of the things that didn’t add value.
“The first principle of Simple by Design is design with Forward Thinking. With that deep customer engagement, anticipate what your customers are going to ask for in the future,” he explained. “Are there things we can do to the design of the product that, without increasing cost, that will allow us to make changes to it at a later date? The second principle is Design to Reduce, so to reduce complexity, can we reduce the number of new parts that we have, can we reduce the number of new suppliers we have? Can we eliminate proprietary materials that might be hard to come by?
“Design to Reuse – can we reuse parts that already exist? Why do we need to invent new when we’ve already got very similar or exactly what we need released into the system…and then finally, if we do the first three, we should see flow in the factory. We should not see the kind of bottlenecks that we experience today.”

Maxwell said that Dassault comes in with software tools that provide access to data, which “is the big game changer.” He talked about all of the many books and catalogs that were in his office at the beginning of his career, noting that engineers today just can look at all of this information online, because they have access to data. Parker Hannifin estimates that it has about 26 million active part numbers, which is a lot to keep track of, and Maxwell said that roughly 45% of a typical design engineer’s time is spent searching for information.
“So if I had access to the data behind that 26 million part numbers, what would happen? And today, I’m not embarrassed to say that generally we don’t. There’s a lot of things that we do many many times, we’re a very diversified company, we’re global, ” Maxwell said. “It’s not unusual for people to spend their entire career here in the company and not talk to a lot of other operating divisions…outside of the one they work in. So what if I could connect them and give them access to information, what kind of leverage might I enjoy?”
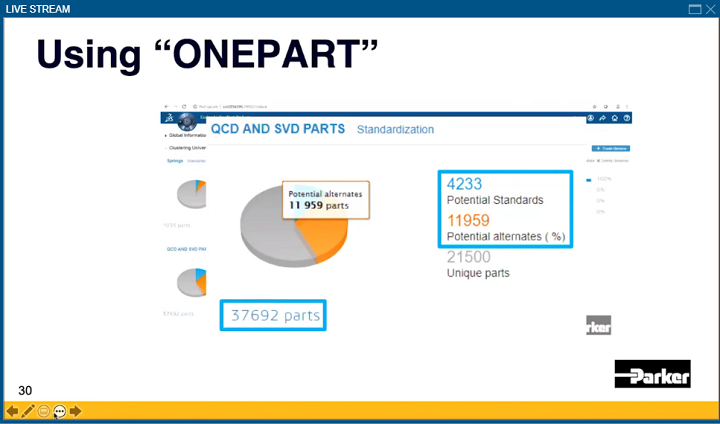
He brought up the company’s usage of Dassault’s EXALEAD OnePart, which can give multiple division access to this kind of information. Maxwell said that this software was used “early on in testing and in value creation,” which was very helpful in finding duplicate parts or component-level parts that already exist in the system, so no one had to create a new part.
Below is a test case he showed of Parker successfully using Dassault tools. FET is an industry-standard 6000 PSI thread to connect couplings, and there are a lot of competitors for parts like this. The company was working to design a new series that was more of a premium product than the original FET.

“We applied simplified design principles,” he explained. “There’s four different sizes, it was bespoke, very distinct from the FET series that was standard. It was fully validated and ready for launch. But it added 147 component parts to the value stream.”
The team focused here, and used the simplified design principles to make the decision to recycle the validated part, and go back to the drawing board.
“Is there an opportunity for us to reuse some of the parts that already exist in the FET series in the new 59 series, but still maintaining the 59 series’ premium features and benefits?”

You can see the results of keeping things simplified above—123 parts were eliminated, while keeping the series at 100% function. The new 59 series shares 90% of its components with the original FET series, and no additional capital was spent on equipment. Costs and inventory went down, and delivery went up, which Maxwell called a “great example of flow.”
Swedberg then introduced Florence Verzelen, Executive Vice President, Industry, Marketing, Global Affairs and Workforce of the Future for Dassault Systèmes, who would discuss “How to Transform the New Normal into an Opportunity.”

She opened by discussing how the COVID-19 crisis has changed everything, such as having to stay home and social distancing, and I’m sure we all agreed with this statement. But now we’re entering a new phase of building back after the pandemic, and building back better, as businesses reopen.
“How do you think you managed during COVID?” she asked. “Are you ready to transform, to perform better in the new normal world? Do you know how to become more resilient and therefore be prepared for the next crisis?”
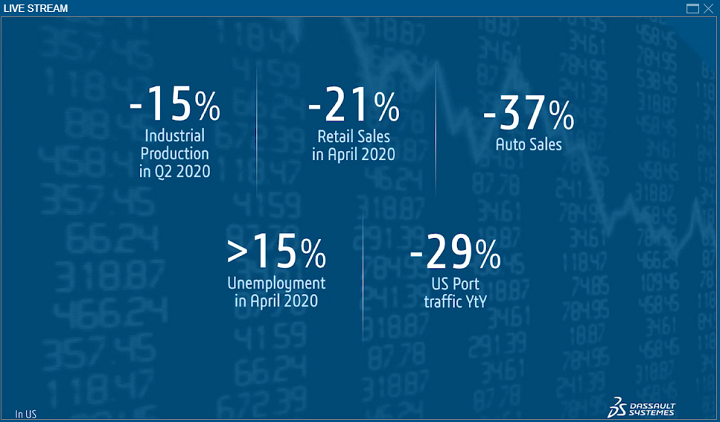
Verzelen discussed some of the stark numbers coming out of the pandemic, such as 53 million—the number of jobs considered to be “at risk” during confinement and quarantine.
“In the 21st century, we have never seen a crisis of this amplitude,” she said. “And when it happens, as industry leaders, there are really two things, two imperatives, we should consider. Ensure the survival of our company, and contribute to the safeguard of the economy.”
There are five actions to take here, and the first priority is to protect employees and make sure they can safely do their jobs.
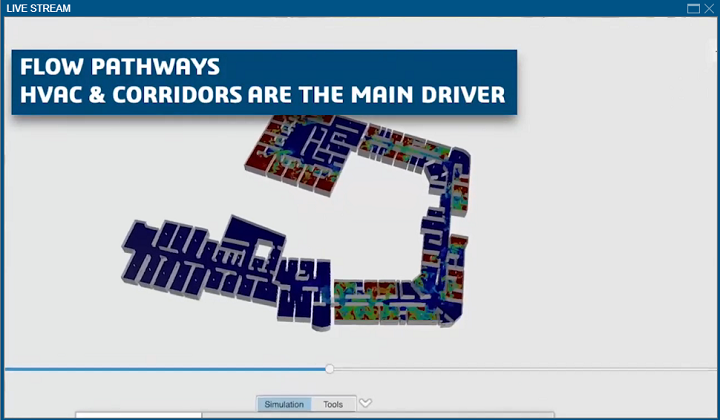
Verzelen explained that the 3DEXPERIENCE tool SIMULIA can help with this in many ways, such as simulating the airflow in a building’s corridors. She also said that companies can “implore their employees to work from home” without disruption, which is possible thanks to Dassault’s cloud solution.
The second thing necessary to keep your company surviving is maintaining its financial health.
“COVID-19 has affected the liquidity of many companies,” she said. “Less revenue, more costs…and in order to make decisions, you need to be able to build a scenario.”
Online sales can help keep companies afloat during a crisis, and also help maintain the connection to customers. Dassault can help with these as well through its data analytics solutions and digital tools. Adapting your company’s marketing and sales for an online experience is the third way to ensure its survival.

The fourth thing is to safeguard the supply chain. The disruption of one supplier can decimate production all the way down the whole chain, which can include suppliers in locations all over the world.
“During a crisis, it becomes essential to know where the weak points are,” Verzelen said. “This again we can do thanks to digitalization and thanks to data analytics.”

Finally, companies need to help the ecosystem, otherwise it will not survive. Dassault made sure that all of its solutions and tools were readily available on the cloud so that all customers could continue to work to keep the ecosystem going.
But, even though the world is slowly coming out of confinement, Verzelen warns that “it’s not over yet.” The use of automation will likely increase, and e-commerce is skyrocketing in Italy.
“It’s the beginning of a new phase. It’s the beginning of what we call the new normal.”

A lot of decisions need to be made when you’re restarting a business. Again, Dassault can help with this by building scenarios, so companies know the right steps to take, and in what order, to successfully reopen.
“We all have to change,” Verzelen said. “We’re developing new capabilities for employees, and making learning experiences available online to make sure your teams are ready. Returning to business probably means we need to rethink our supply chain, and we know that a contact-limited economy is here to stay. So you should push for e-commerce, and be prepared to work in contact-limited economy.”

She stated that the 3DEXPERIENCE allows companies to “unlock unlimited value,” and help us cope during this new normal.
“There are many ways to be resilient, and all of those ways are linked to innovation and sustainability.”
The paradigm has changed, and we need to be realistic going forward, and focus on sustainability in operations and business models, such as turning to additive manufacturing if your usual supplier can’t get you what you need in time.
“With the 3DEXPERIENCE platform you can create this kind of business model…create more efficiently, design more quickly,” she said.
“In a nutshell, we are going through very difficult times right now…But this crisis can also be seen as an opportunity to rethink what we do, and build back better.”
Finally, Swedberg introduced three additional Dassault panelists for the final discussion: Dr. Ales Alajbegovic, Vice President, SIMULIA Industry Process Success & Services; Garth Coleman, Vice President, ENOVIA Advocacy Marketing; and Eric Green, DELMIA’s Brand Marketing Vice President. These three are in charge of the content for the rest of 3DEXPERIENCE: A Virtual Journey, as it continues on:
- “Fueling Innovation for the New Agile Enterprise,” August 26th
- “Modeling & Simulation, Additive Manufacturing,” September 23rd
- “Enabling Business Continuity Using the Cloud,” October 14th
L-R: Swedberg, Green, Coleman, Alajbegovic
Green said that three themes would be articulated in these upcoming sessions, all of which will fall under the “sustainable operations” umbrella: data-driven decision-making, leveraging agile success and being agile for success, and business resiliency. Coleman mentioned that the many customer references and testimonials found on the 3DEXPERIENCE site provide many examples of how the platform has helped customers innovate across every industry…even wine-making! Dr. Alajbegovic said that they are “very excited” about the upcoming modeling and simulation sessions and additive manufacturing panels.
“In our sessions, we will look at ways to enable the marriage between modeling and simulation, thus revolutionizing design,” Dr. Alajbegovic said.
It’s not too late to register for 3DEXPERIENCE: A Virtual Journey, so sign up today to enjoy access to further digital programming from Dassault Systèmes.
The post 3DEXPERIENCE: A Virtual Journey, Part 1 appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.