We’ve got virtual events and webinars this week covering everything from sustainability and forming to metal and medical additive manufacturing. Read on to learn what’s available!
NatureWorks 3D Considers Sustainability in AM

Biotechnology company NatureWorks 3D is hosting a webinar this Tuesday, August 18th, at 1 pm EDT, titled “Printing Consciously: Considering Sustainability in 3D Printing.” The free webinar will last about one hour, and cover topics such as circular vs. linear model of materials, mechanical and chemical recycling, best practices for used FFF 3D printing materials, environmental impacts of using bio-based and petrochemical-based filaments, and more. Dan Sawyer, the company’s Business Development Manager, and Deepak Venkatraman, Applications Development Engineer for NatureWorks, will share some thoughts and insights into how polymers fit into the circular economy approach in order to decrease the AM industry’s impact on the world.
“A renewed focus on climate change and the impacts petrochemical plastics have on the environment has many individuals and companies considering how they can incorporate more sustainable practices into their efforts. The additive manufacturing industry has long been a leader in how technology can fit into a progression toward a more sustainable production. In this webinar, we will dig into the sustainability attributes behind the materials often used in fused filament fabrication (FFF) processes that have an environmental impact. We’ll also talk about how 3D prints fit within common waste scenarios as well as new sustainability frameworks like the circular economy.”
There will be a question and answer session at the end of the webinar; register here to attend.
EOS Introduces the INTEGRA P 450

Also on August 18th, EOS is holding a webinar at 2 pm EDT to introduce its latest system, titled “From R&D to Production: Introducing the INTEGRA P 450.” This mid-size, SLS industrial additive manufacturing system was developed by EOS North America, based off of direct feedback from the manufacturing community and built to “meet the demand for additive manufacturing of polymers, it empowers designers, production engineers and material makers alike.” In addition to gaining an understanding of the INTEGRA P 450‘s material compatibilities and development opportunities, attendees will also learn about the company’s new open software platform. Speakers will be Fabian Krauss, EOS North America’s Global Business Development Manager, Polymers; Mohit Chaudhary, Additive Manufacturing Specialist, Polymers – Solution Engineering, for EOS North America; and Mike Conner, EOS North America’s Vice President of Service and Support.
“Discover how the INTEGRA P 450 is truly the most flexible and accommodating SLS industrial 3D printer on the market, with an impressive array of new user-friendly features that offer unprecedented productivity, material compatibility, and simple serviceability.”
Register for the webinar here.
Protolabs Discussing Forming and Formed Features

As part of its ongoing webinar series, Protolabs will be discussing sheet metal forming during its webinar, “A Deep Dive on Forming and Formed Features,” on Wednesday, August 19th, at 2 pm EDT. James Hayes, Protolabs Applications Engineer and the company’s technical applications engineering expert for sheet metal fabrication, will offer insight into forming techniques and equipment, as well as important design considerations for sheet metal forming, and how they can impact part geometry. You’ll leave with new knowledge and insight into how to leverage formed features, and improve sheet metal part designs.
“Understanding the ins and outs of sheet metal forming can be fraught with challenges, however there are some important things to know that can result in better designed, more cost-efficient parts. In addition, considerations between how different formed features can impact your product throughout its’ lifecycle can help you achieve your product goals and bring your ideas to market at record speeds.”
Register for the webinar here.
ASME’s AM Medical Live Webinar

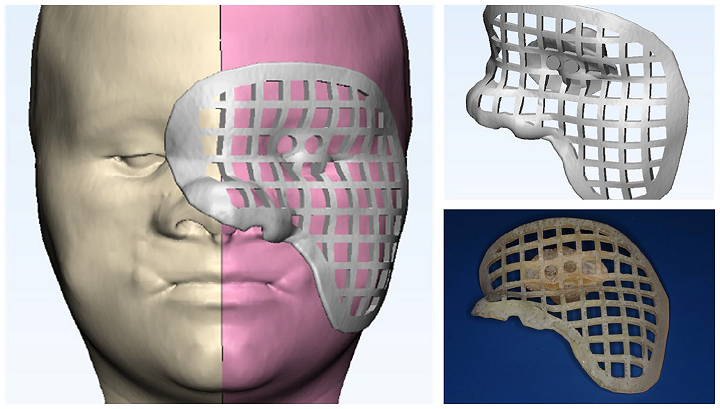
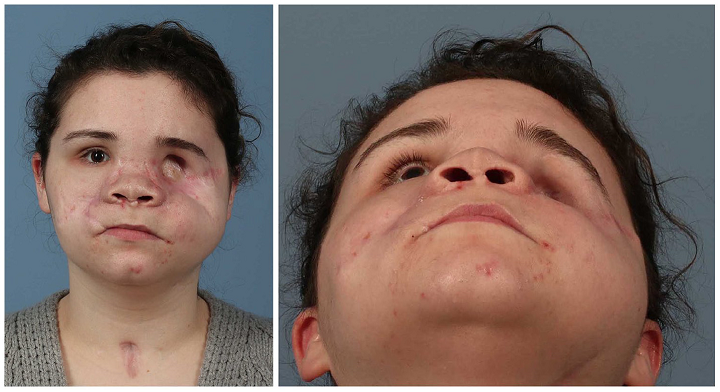
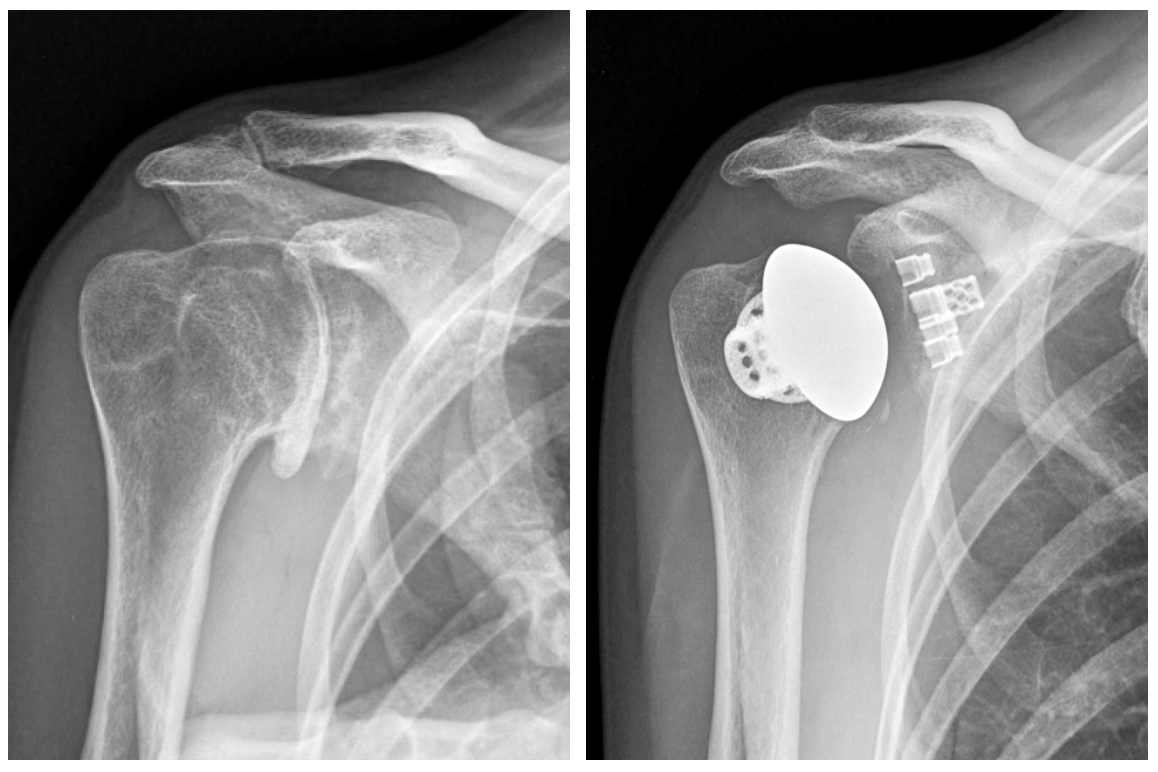
Last week, ASME was powering the AM Industry Summit, for 3D printing professionals working in the aerospace and defense and medical device manufacturing fields. Now it’s hosting a live webinar this Thursday, August 20th, from 2-3 pm EDT, supported by Women in 3D Printing and titled “Integrating 3D Printing with Other Technologies at the Point of Care.” Speakers will be Sarah Flora, the Radiology Program Director for the 3D Lab at Geisinger Health; Amy Alexander, MS, Senior Biomedical Engineer at the Mayo Clinic’s Anatomic Modeling Lab; and the Director of the 3D Imaging Lab at Montefiore Medical Center, Nicole Wake, PhD. They will be discussing how 3D printing is often a very important medical tool when it comes to patient care.
“Whether anatomical models or guides are used for education or surgical planning, radiologists, surgeons, and engineers work together to improve the patient experience. Leveraging 3D printing with other technologies can expand the value within a clinical setting. Three leading clinical engineers will discuss technologies that can be used together to extend the usefulness of 3D printing including silicone casting, surface scanning, augmented reality, and more. Join the discussion to explore the unexpected ways to increase the benefits of 3D printing.”
The webinar is free to attend, and you can register for it here.
IDTechEx on Metal Additive Manufacturing

Finally, also on August 20th, IDTechEx will be holding its latest free, expert-led webinar, “Metal AM: Short-Term Pain, Long-Term Gain.” Presented by Dr. Richard Collins, IDTechEx’s Principal Analyst, the webinar, which shares some research from the company’s detailed “Metal Additive Manufacturing 2020-2030” report, will provide an overview of the latest key trends and market forecast for metal additive manufacturing, the latest material considerations and entrant analysis, technology benchmarking, the impact of COVID-19, and more.
“Metal additive manufacturing has been gaining traction. Increased number of use-cases, end-users progressing along the learning curve, more competition, and a maturing supply chain. The applications have been led in high-value industries most notably aerospace & defence and medical, many more are emerging in automotive, oil & gas, and beyond. These sectors have had very different fates during the global pandemic and the knock-on effect will be profound. There are some silver-linings and the long-term outlook is positive for this industry, but it will not be an easy ride. IDTechEx forecast the total annual market for metal additive manufacturing to exceed $10bn by 2030. This is not before a very challenging immediate future; a result of the COVID-19 pandemic.”
Three different sessions of this 30-minute webinar will be offered, the first of which will actually take place at 9 pm EST, on the 19th. The next one will be at 5 am EST, and the final session will be at 12 PM EST. You can register for your preferred session here.
ASTM’s AM General Personnel Certificate Program

Don’t forget, the ASTM International Additive Manufacturing Center of Excellence (AM CoE) is still offering its online AM General Personnel Certificate course, which continues through August 27th and is made up of eight modules covering all the general concepts of the AM process chain. Register for the class here.
Will you attend any of these events and webinars, or have news to share about future ones? Let us know!
The post 3D Printing Webinar and Virtual Event Roundup, August 16, 2020 appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.