Managing bone defects and injuries using traditional treatments can be slow and expensive. When bones break, bone cells can usually repair them, unless the break is too large. In that case, clinicians have historically turned to bone grafts using non-essential segments of bone taken from other parts of the same patient; bone from the hips, pelvis, chin, or ribs can do the job. Unfortunately, this requires additional surgeries, which translates into more pain for patients, and basically, there is a limit to how much non-essential bone surgeons can take from a patient.
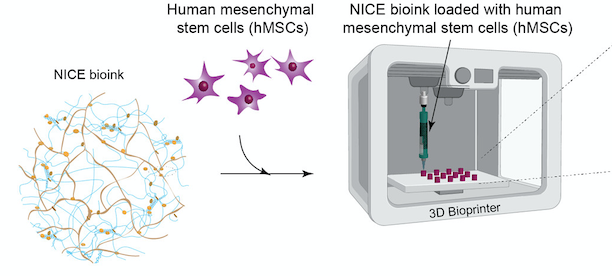
As in many fields, bioprinting is disrupting the way healthcare specialists think about solving problems. So with an estimated 500,000 annual bone grafting procedures in the United States and more than 2 million around the world, an efficient bone substitute could change millions of lives. In a search to fabricate patient-specific, implantable 3D constructs for regenerative medicine, scientists in the Department of Biomedical Engineering at Texas A&M University have developed a new bioink formulation for 3D bone bioprinting called NICE, which is short for Nanoengineered Ionic–Covalent Entanglement, and have gone on to demonstrate that this bioink can precisely reconstruct large bone structures based upon CT scans that were obtained from actual patients.
Led by Akhilesh K. Gaharwar, an associate professor in the Department of Biomedical Engineering, the research group developed a highly printable bioink as a platform to generate anatomical-scale functional tissues. Their study was recently published in the American Chemical Society’s Applied Materials and Interfaces scientific journal, whereby they state that the NICE bioinks allow precise control over printability, mechanical properties, and degradation characteristics, enabling custom 3D fabrication of mechanically resilient, cellularized structures.
Bioprinting requires cell-laden biomaterials that can flow through a nozzle like a liquid, but solidify almost as soon as they’re deposited. This is why bioinks need to act as both cell carriers and structural components, requiring them to be highly printable while providing a robust and cell‐friendly microenvironment. However, the research group realized that many current bioinks lack sufficient biocompatibility, printability, structural stability, and tissue‐specific functions needed to translate this technology to preclinical and clinal applications.
To address this issue, Gaharwar and his team are leading efforts in developing the more advanced NICE bioinks, essentially a combination of two reinforcement approaches, ionic-covalent entanglement, and nanoreinforcement. In fact, the researchers claim that to design the NICE reinforced bioinks for osteogenic tissue bioprinting, the bioink must be highly printable, mechanically strong, induce osteogenic differentiation, and be biodegradable. However, the difficulty of combining these requirements into a single bioink has been a major obstacle in bioprinting since its inception. So by combining these two distinct reinforcement methods, NICE becomes a robust and superior bioink while providing a highly hydrated and cell-friendly microenvironment for bone bioprinting.

NICE printed structures are highly flexible and resilient, as seen in these 3D printed tube structures that can be completely collapsed and quickly regain their shape (Credit: Texas A&M Engineering)
According to Texas A&M Today, Gaharwar said that developing replacement bone tissues could create exciting new treatments for patients suffering from arthritis, bone fractures, dental infections, and craniofacial defects.
“The next milestone in 3D bioprinting is the maturation of bioprinted constructs toward the generation of functional tissues,” Gaharwar said. “Our study demonstrates that NICE bioink developed in our lab can be used to engineer 3D-functional bone tissues.”
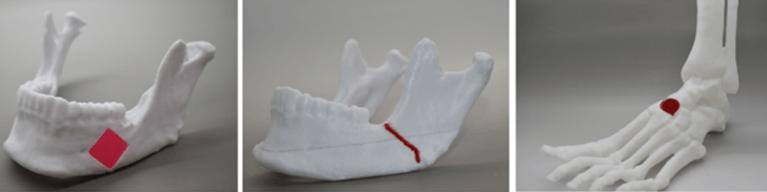
To illustrate the practical utility of NICE bioinks for bone tissue reconstruction, the team demonstrated how to create full-scale bioprinted implants customized for craniofacial defects on real patient CT scans. Relying entirely on open-source software, they used the free 3D modeling software Meshmixer to process the models and create bone defects, and the 3D printing applications PrusaSlicer and Repetier Host to bioprint the scaffolds. After bioprinting, the scaffold was crosslinked and implanted in a thermoplastic model of the lower jaw to demonstrate the closeness of fit. Strength of fit was also demonstrated by injecting and crosslinking NICE bioink between two sections of a full-thickness fracture to prove that NICE is able to quickly adhere surfaces together and resist shearing and delamination forces.
Funded by the National Institutes of Health (NIH)’s Director’s New Innovator Award, a National Science Foundation (NSF)’s Award and an X-Grant from Texas A&M University, the researchers suggest they have discovered a new way to design and produce 3D bioprinted bone tissue to benefit bone regeneration.
Moreover, Gaharwar claims to have demonstrated that the highly printable NICE bioinks can precisely reconstruct large bone structures from CT scans obtained from actual patients. The aim of the research is to enable patient-specific bioprinting of bone scaffolds to precisely match their injuries. The researchers stated their desire to have this technique act as a customizable and easy to work with an alternative to autografts that will provide surgeons with greater options for bone surgery. And with the ultimate goal of getting NICE bioink technology from bench to bedside, Gaharwar’s team plans to establish the in vivo functionality of the 3D bioprinted bone tissue.
The post Texas A&M Researchers Make “NICE” Bioinks to Create Functional Bone Tissues appeared first on 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing.